You know the mechanism of HBr addition to alkenes, and you know the effects of various substituent
Question:
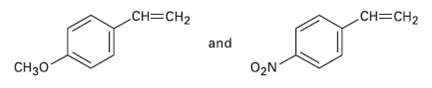
You know the mechanism of HBr addition to alkenes, and you know the effects of various substituent groups on aromatic substitution. Use this knowledge to predict which of the following two alkenes reacts faster with HBr. Explain your answer by drawing resonance structures of the carbocationintermediates.
Transcribed Image Text:
CH=CH2 CH=CH2 and CH30 O2N
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 64% (14 reviews)
Problem shows the mechanism of the addition of HBr to 1phenylp...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
By drawing resonance structures for the carbocation intermediates, show why aromatic substitution in pyridine - N - oxide occurs at the 4 - position rather than at the 3 - position.
-
Predict which of the following two compounds will undergo an E2 reaction more rapidly: CI
-
The addition of HBr to which of the following alkenes is more highly regioselective? a. b. CH CH CH,CHC=CH, CH 3C CHCH or - CH CH3 or
-
How many objects are eligible for garbage collection at the end of the main() method? A. None. B. One. C. Two. D. Three. E. The code does not compile. F. None of the above. package store; public...
-
Determine how many personal and dependency exemptions would be available in each of the following independent situations. Specify whether any such exemptions would come under the qualifying child or...
-
Describe the advantages of an experiment using a crossover design instead of independent samples to compare the tanning methods.
-
Hiker Company completes the following transactions during the current year. May 9 Purchases 200 shares of Higo stock as a short-term investment in available-for-sale securities at a cost of $25 per...
-
You are the vice president of International InfoXchange, headquartered in Chicago, Illinois. All shareholders of the firm live in the United States. Earlier this month you obtained a loan of 5...
-
In its 2024 income statement, Tow Inc. reported proceeds from an officer's life insurance policy of $90,000 and depreciation of $250,000. Tow was the owner and beneficiary of the life insurance on...
-
The following transactions apply to Jova Company for Year 1, the first year of operation: 1. Issued $10,000 of common stock for cash. 2. Recognized $210,000 of service revenue earned on account. 3....
-
How would you synthesize the following compounds from benzene? Assume that ortho and Para isomers can beseparated. (b) CH2CHCH3 (a) CH Br O2N SO3H
-
Benzyl bromide is converted into benzaldehyde by heating in dimethyl sulfoxide. Propose a structure for the intermediate, and show the mechanisms of the two steps in thereaction. CH2BR E2 reaction...
-
Briefly describe how repeaters, hubs, switches, and routers differ from one another.
-
Pink Jeep Tours offers off-road tours to individuals and groups visiting the Southwestern U.S. hotspots of Sedona, Arizona, and Las Vegas, Nevada. Take a tour of the companys Web site at...
-
The following are unrelated accounting practices: 1. Pine Company purchased a new \(\$ 30\) snow shovel that is expected to last six years. The shovel is used to clear the firm's front steps during...
-
Identify whether the following statements are true or false. 1. One argument for IFRS is that there is less globalization in the world. 2. IFRS is accepted as GAAP in every country of the world. 3....
-
You will need isometric dot paper for this question. Part of a pattern using four rhombuses is drawn on isometric dot paper below. By drawing two more rhombuses, complete the pattern so that it has a...
-
Fred Flores operates a golf driving range. For each of the following financial items related to his business, indicate the financial statement (or statements) in which the item would be reported:...
-
WHAT ARE THE COMPONENTS OF A LAN?
-
This problem continues the Draper Consulting, Inc., situation from Problem 12-45 of Chapter 12. In October, Draper has the following transactions related to its common shares: Oct 1 Draper...
-
Forest cabins, remote mobile homes, Amish farms, and residential structures in locations where electricity is not available are often equipped with absorption refrigerators that rely on changes from...
-
Draw the structures for these compounds: (a) 1, 6-Hexanedioic (b) Ethyl 2-ethyl-2-hydroxybutanoate (c) 2-Amino-30syslohexyl-1-propanol (d) tert-Butyl 2-hydroxy-5-octenoate (e) N, N,...
-
Draw structures for these compounds: (a) 3-Methoxybenzoic acid (b) p-Phenyl phenol (c) 2-Methylbutyl p-toluenesulfonate (d) 4-Cyano-3. 5-dimethyloctanal (e) Sodium acetate (f) 2, 4-Dinitrotoluene (g)...
-
Provide names for these compounds: b) a) , CO.H c) CH,0 d) NH2
-
3 . Accounting.. How does depreciation impact financial statements, and what are the different methods of depreciation?
-
NEED THIS EXCEL TABLE ASAP PLEASE!!!! Presupuesto Operacional y C lculo del COGS Ventas Proyectadas: Ventas Proyectadas: $ 4 5 0 , 0 0 0 Precio por unidad: $ 4 5 0 Unidades vendidas: 4 5 0 , 0 0 0 4...
-
The wash sale rules apply to disallow a loss on a sale of securities_______? Only when the taxpayer acquires substantially identical securities within 30 days before the sale Only when the taxpayer...

Study smarter with the SolutionInn App


