Question: 10-Bromo--chamigrene, a compound isolated from marine algae, is thought to be biosynthesized from -bisabolene by the following route: Draw the structures of the intermediate bromonium
10-Bromo-α-chamigrene, a compound isolated from marine algae, is thought to be biosynthesized from γ-bisabolene by the following route: Draw the structures of the intermediate bromonium and cyclic carbocation, and propose mechanisms for all three steps.
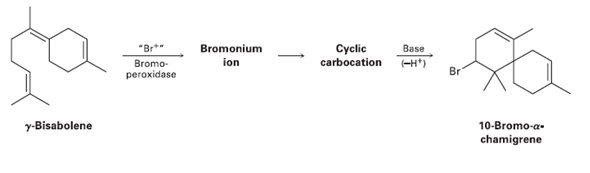
Cyclic carbocation -H*) Bromonium "Br*" Bromo- Base ion Br peroxidase y-Bisabolene 10-Bromo-a- chamigrene
Step by Step Solution
3.22 Rating (166 Votes )
There are 3 Steps involved in it
Br Br Bromo peroxid... View full answer

Get step-by-step solutions from verified subject matter experts
Document Format (1 attachment)
22-C-OC-A (174).docx
120 KBs Word File


