A block of mass m 1 = 2 kg slides along a frictionless table with a speed
Question:
A block of mass m1 = 2 kg slides along a frictionless table with a speed of 10 m/s. Directly in front of it, and moving in the same direction with a speed of 3 m/s, is a block of mass m2 = 5 kg. A massless spring with spring constant k = 1120 N/m is attached to the second block as in Figure
(a) Before m1 runs into the spring, what is the velocity of the center of mass of the system?
(b) After the collision, the spring is compressed by a maximum amount Δx. What is the value of Δx?
(c) The blocks will eventually separate again. What are the final velocities of the two blocks measured in the reference frame of the table?
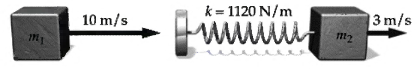
k = 1120 N/m wwww 3 m/s 10 m/s m2
Step by Step Answer:
a We use Equation 813 v cm 2 10 5 37 ms 5 ms b At max Compression u 1 u 2 0 K K cm K c...View the full answer



Fundamentals of Ethics for Scientists and Engineers
ISBN: 978-0195134889
1st Edition
Authors: Edmund G. Seebauer, Robert L. Barry
Students also viewed these Mechanics questions
-
A block of mass m1 = 3.70 kg on a frictionless plane inclined at angle = 30.0o is connected by a cord over a mass less, frictionless pulley to a second block of mass m2 = 2.30 kg (Figure). What are?...
-
A block of mass m1 = 2.00 kg and a block of mass m2 = 6.00 kg are connected by a mass less string over a pulley in the shape of a solid disk having radius R = 0.250 m and mass M = 10.0 kg. These...
-
In Figure (a) a block of mass m lies on a horizontal frictionless surface and is attached to one end of a horizontal spring (spring constant k) whose other end is fixed. The block is initially at...
-
Suppose you buy Bond A at time=0. It is a 2-year, risk-free bond with coupon rate of 7% that compounds annually, and it has a face value of $1,000. (3 points) a) At time = 0, suppose you buy Bond A...
-
What does a negative value for AFN mean?
-
Fermat, Inc., has acquired two new companies, one in consumer products and the other in financial services. Fermat's top management believes that the executives of the two newly acquired companies...
-
Calculate the standard Gibbs free energy change and the equilibrium constant at 1 bar and \(298.15 \mathrm{~K}\) for the ammonia synthesis reaction \[ \mathrm{C}_{2} \mathrm{H}_{5}...
-
Part 1. Meir, Benson, and Lau are partners and share income and loss in a 3:2:5 ratio. The partnerships capital balances are as follows: Meir, $ 168,000; Benson, $ 138,000; and Lau, $ 294,000. Benson...
-
If f(x) = +3t + 2) dt +2)dt then f"(x)=
-
You are the management accountant of T plc. The following computer printout shows details relating to April: T plc uses a standard absorption costing system. There was no opening or closing work in...
-
Night after night, Lucy is tormented by nocturnal wailing from the house next door. One day she seizes a revolver, stalks to the neighbors window with a crazed look in her eye, takes aim, and fires a...
-
A bullet of mass m is fired vertically from below into a block of wood of mass M that is initially at rest, supported by a thin sheet of paper. The bullet blasts through the block, which rises to a...
-
Longhorn Corporation provides low-cost food delivery services to senior citizens. At the end of the year, the company reports the following amounts: In addition, the company had common stock of...
-
Why are the value and cash flows of levered equity less than if the firm had issued unlevered equity?
-
Why will a short-term and long-term investor with the same beliefs be willing to pay the same price for a stock?
-
Why are investors indifferent to the firms capital structure choice?
-
How can you estimate a firms stock price based on its projected free cash flows?
-
Should uninformed investors expect to make money by trading based on news announcements?
-
Recognize a multiple regression model and be able to interpret the coefficients in that model.
-
What mass of KBr (in grams) should you use to make 350.0 mL of a 1.30 M KBr solution?
-
Show that for some fuel cells the efficiency value, e = r G/ r H,can have a value greater than 1.00. Can you identify one such reaction? Use data from Appendix d. TABLE D.1 Ground-State Electron...
-
A vertical parallel-plate capacitor is half filled with a dielectric for which the dielectric constant is 2.00 (Fig. P26.70a). When this capacitor is positioned horizontally, what fraction of it...
-
Capacitors C1 = 6.00 F and C2 = 2.00 F are charged as a parallel combination across a 250-V battery. The capacitors are disconnected from the battery and from each other. They are then connected...
-
Calculate the equivalent capacitance between the points a and b in Figure P26.72. Note that this is not a simple series or parallel combination. (Suggestion: Assume a potential difference V between...
-
This is a slip and fall case. Plaintiff was injured when she slipped and fell in the defendant's fast food restaurant. Plaintiff claims the floor was wet and slippery when she fell. Defendant denies...
-
1. Explain what the "Deinstitutionalization Mandate" is. This should be done in at least two paragraphs. 2. In another two paragraphs, discuss how status offenses are connected to the...
-
You are the owner of a very small business that sells gourmet coffee. You sell only one product, a 12-ounce bag of whole-bean French roast coffee. You sell each bag of coffee for $14 each, but due to...

Study smarter with the SolutionInn App


