A carbanion is a species that contains a negatively charged, trivalent carbon. (a) What is the electronic
Question:
A carbanion is a species that contains a negatively charged, trivalent carbon.
(a) What is the electronic relationship between a carbanion and a trivalent nitrogen compound such as NH3?
(b) How many valence electrons does the negatively charged carbon atom have?
(c) What hybridization do you expect this carbon atom to have?
(d) What geometry is the carbanion likely tohave?
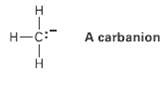
Transcribed Image Text:
H-C: A carbanion
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 55% (9 reviews)
H H H H H H a A carbanion is isoelectronic with h...View the full answer

Answered By

Sachin Kumar
1. Working on “Enhancing Anthocyanin Production in Tomato”.
2. Worked in TB C&DST Lab of Dr. S.N. Medical College as an intern for 4 weeks, on the topic
“Detection of Rif resistant in presumptive TB samples using Cartridge based nucleic acid
amplification test; A hospital-based study” and worked in BSL 3 Lab & Equipment.
3. Published research paper on "Study effects of different media on the production of siderophore by certain microorganisms".
4. Published research paper on "Implementing the Physical Web Using Bluetooth Low Energy Based Beacons and a Mobile App".
5. TECHNICAL SKILLS AND INSTRUMENT HANDLING
? Microbiological Techniques: Microscopy, micrometry, staining, isolation of pure culture, genomic DNA isolation, human body microflora analysis, drug sensitivity of microbes, etc.
? Immunological techniques: ABO blood grouping system, Rh-factor determination, Widal test, serum extraction, disc diffusion test, etc.
? Instruments well versed with LAF cabinet, centrifuge, electrophoresis, Autoclave, etc.
? Biosafety Levels: BSL1, BSL2, BSL3.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
How many valence electrons does carbon have?
-
How many valence electrons does each of the following atoms have? (a) Na (b) Cl (c) Si (d) B (e) Ne (f) N
-
How many valence electrons does the each of the following dietary trace elements have? (a) Zinc (b) Iodine (c) Silicon (d) Iron
-
People who earn a higher salary can afford more goods, including health care. However, according to Grossman, they will choose a higher desired health stock. Why is this so, according to the model?
-
Amber Industries (a sole proprietorship) sold three 1231 assets during 2016. Data on these property dispositions are as follows: a. Determine the amount and the character of the recognized gain or...
-
Spike recovery and detection limit. Species of arsenic found in drinking water include AsO3 3 - (arsenite), AsO43- (arsenate), (CH3)2AsO23- (dimethylarsinate), and (CH3)AsO32- (methylarsonate) Pure...
-
If the observed speed of sound in steel is \(5300 \mathrm{~m} / \mathrm{s}\), determine the bulk modulus of elasticity of steel in \(\mathrm{N} / \mathrm{m}^{3}\). The density of steel is nominally...
-
A contract made for the sale of a farm stated that the buyers deposit would be returned if for any reason the farm cannot be sold. The seller later stated that she had changed her mind and would not...
-
a. briefly mention three types of audience. b. Consider an airline company with its employees including crews and pilots who offer services to the customers and passengers. Please explain...
-
Shefford Cutlery extends a lifetime replacement warranty on all units sold. Using past experience, the company estimates that 0.5% of units sold will be returned and require replacement at an average...
-
Almost all stable organic species have tetravalent carbon atoms, but species with trivalent carbon atoms also exist. Carbocations are one such class of compounds. (a) How many valence electrons do...
-
Divalent carbon species called carbenes care capable of fleeting existence. For example, methylene: CH2, is the simplest carbene. The two unshared electrons in methylene can be either spin-paired in...
-
The processing cost of making a payment through the ACH system is roughly half the cost of making the same payment by check. Why, therefore, do firms often rationally choose to make payments by check?
-
why is cognitive psychology so interesting? what does perception in cognitive psychology tell you? What is compelling and surprising about consciousness and attention in cognitive psychology? how can...
-
Why would a company consider increasing the amounts that employees have to pay towards their benefits by increasing premiums or deductibles?
-
Define cognitive psychology and identify key milestones in the development of cognitive psychology as a discipline?
-
What is the biggest concern for cognitive psychology? 2. How is cognitive psychology ethical? 3. What processes of cognitive psychology support the development of creativity?
-
What is Cognitive Psychology? What are 3 ways that Cognitive Psychology influences the practice of psychology today?
-
Which of the following investment vehicles provide investors with the highest degree of liquidity? A. Open-end funds B. Private equity funds C. Limited partnerships
-
What are the principal alloying elements in SAE 4340 steel?
-
At a subcritical temperature, the branch of the PengRobinson equation of state for V > b exhibits a van der Waals loop. However, there is also interesting behavior of the equation in the ranges V < b...
-
Provide the values for the missing K a or p K a in the following examples: (a) p K a = 4; K a =? (b) K a = 1 x 10 16 ; p p K a =? (c) p K a = 38; K a =? (d) K a = 1 x 10 6 ; p K a =?
-
Indicate whether these compounds are weaker or stronger acids than water (the K a for water is 1.8 x 10 ?16 ; the p K a is 15.74): (K=100) a) HCIO4 c) HOCOH (PK = 6.35) b) HCCH (pK = 25) d) CH3CH...
-
Indicate whether these species are weaker or stronger bases than hydroxide ion. The Ka is or pKa values are for the conjugate acids. a) :NH, (K = 10-38) c) NH, (PK, = 9.24) b) CHCHCH d) :CI: (pK =...
-
Generous Limited has issued a callable bond at 5.35% coupon with 14 years left to maturity can be called in 4 years. The call premium is one year of coupon payments. It is offered for sale at...
-
If the nominal interest rate is 14%, and inflation is 4%, what is the real interest rate? 22.A bond investment yielded 8%. If inflation was 3%, what real return did the bond offer? 23.The following...
-
Bierce Corporation has two manufacturing departments--Machining and Finishing. The company used the following data at the beginning of the year to calculate predetermined overhead rates: Estimated...

Study smarter with the SolutionInn App


