Question: Indicate whether these species are weaker or stronger bases than hydroxide ion. The Ka is or pKa values are for the conjugate acids. a) :NH,
Indicate whether these species are weaker or stronger bases than hydroxide ion. The Ka is or pKa values are for the conjugate acids.
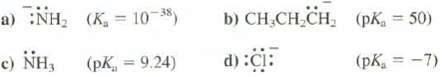
a) :NH, (K = 10-38) c) NH, (PK, = 9.24) b) CHCHCH d) :CI: (pK = 50) (pK = -7)
Step by Step Solution
3.34 Rating (160 Votes )
There are 3 Steps involved in it
The weaker the acid the stronger its conjugate base if the ... View full answer

Get step-by-step solutions from verified subject matter experts
Document Format (1 attachment)
15-C-O-A-B-R (8).docx
120 KBs Word File


