Addition of HC1 to 1-methoxycyclohexene yields 1-chloro-1-methoxycyclo- hexane as the sole product. Use resonance structures to explain
Question:
Addition of HC1 to 1-methoxycyclohexene yields 1-chloro-1-methoxycyclo- hexane as the sole product. Use resonance structures to explain why none of the other regioisomer isformed.
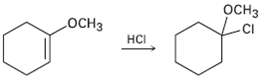
Transcribed Image Text:
оснз оСнз HCI CI CI
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 28% (7 reviews)
ficos OCH 3 H OCH3 Conjugation with the oxygen lone pair electrons makes the double bond more nucl...View the full answer

Answered By

Ashington Waweru
I am a lecturer, research writer and also a qualified financial analyst and accountant. I am qualified and articulate in many disciplines including English, Accounting, Finance, Quantitative spreadsheet analysis, Economics, and Statistics. I am an expert with sixteen years of experience in online industry-related work. I have a master's in business administration and a bachelor’s degree in education, accounting, and economics options.
I am a writer and proofreading expert with sixteen years of experience in online writing, proofreading, and text editing. I have vast knowledge and experience in writing techniques and styles such as APA, ASA, MLA, Chicago, Turabian, IEEE, and many others.
I am also an online blogger and research writer with sixteen years of writing and proofreading articles and reports. I have written many scripts and articles for blogs, and I also specialize in search engine
I have sixteen years of experience in Excel data entry, Excel data analysis, R-studio quantitative analysis, SPSS quantitative analysis, research writing, and proofreading articles and reports. I will deliver the highest quality online and offline Excel, R, SPSS, and other spreadsheet solutions within your operational deadlines. I have also compiled many original Excel quantitative and text spreadsheets which solve client’s problems in my research writing career.
I have extensive enterprise resource planning accounting, financial modeling, financial reporting, and company analysis: customer relationship management, enterprise resource planning, financial accounting projects, and corporate finance.
I am articulate in psychology, engineering, nursing, counseling, project management, accounting, finance, quantitative spreadsheet analysis, statistical and economic analysis, among many other industry fields and academic disciplines. I work to solve problems and provide accurate and credible solutions and research reports in all industries in the global economy.
I have taught and conducted masters and Ph.D. thesis research for specialists in Quantitative finance, Financial Accounting, Actuarial science, Macroeconomics, Microeconomics, Risk Management, Managerial Economics, Engineering Economics, Financial economics, Taxation and many other disciplines including water engineering, psychology, e-commerce, mechanical engineering, leadership and many others.
I have developed many courses on online websites like Teachable and Thinkific. I also developed an accounting reporting automation software project for Utafiti sacco located at ILRI Uthiru Kenya when I was working there in year 2001.
I am a mature, self-motivated worker who delivers high-quality, on-time reports which solve client’s problems accurately.
I have written many academic and professional industry research papers and tutored many clients from college to university undergraduate, master's and Ph.D. students, and corporate professionals. I anticipate your hiring me.
I know I will deliver the highest quality work you will find anywhere to award me your project work. Please note that I am looking for a long-term work relationship with you. I look forward to you delivering the best service to you.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Addition of HC1 to 1, 2-dimethylcyclohexene yields a mixture of two products. Show the stereochemistry of each, and explain why a mixture is formed.
-
Use resonance structures to help you identify all sites of low electron density (δ+) in the following compound:
-
Use resonance formulas to explain why polyacetylene has delocalized molecular orbitals extending over the length of the molecule, whereas the following molecule does not. HHHHH
-
Pipestone Ltd. (Pipestone) uses an aging schedule to estimate the amount of receivables that won't be collected. Pipestone allows its customers up to 60 days to pay amounts owed. Any receivable...
-
Why can't car dealerships simply keep paying based on past systems?
-
Obtain a copy of PCAOB Release No. 104-2016-142. a. What is this PCAOB Release? b. How many issuer audits did the PCAOB review? c. How many issuer audits with audit deficiencies are included in the...
-
There is a market for bets on the outcome of a coin toss. The possible outcomes are heads, tails, and edge. There are three assets traded in that market: Asset A pays \(\$ 1\) independent of the...
-
A company's marketing and accounting departments have determined that if the company markets its newly developed product, the contribution of the product to the firm's profit during the next 6 months...
-
You have a computer system where a customer wants the device to be changed from being a magnetic hard drive to an SSD using a M.2 interface. The desktop computer does not have a M.2 slot. Is what the...
-
Create common size income statements and perform a basic ratio analysis of the two companies for fiscal year 2021, which is the fiscal year ended on Jan. 29, 2022/Jan. 28, 2022. (Note: fiscal year...
-
Myrcene, C10H16, is found in oil of bay leaves and is isomeric with ?-Ocimene (Problem 14.48). It has an ultraviolet absorption at 226 nm and can be catalytically hydrogenated to yield 2,...
-
Hydrocarbon A, C 10 H 14 , has a UV absorption at ? max = 236 nm and gives hydrocarbon B, C 10 H 18 , on catalytic hydrogenation. Ozonolysis of A followed by zinc/acetic acid treatment yields the...
-
A rope is attached from a truck to a 1400-kg car. The rope will break if the tension is greater than 2500 N. Ignoring friction, what is the maximum possible acceleration of the truck if the rope does...
-
1. Which of the following creates a tuple? A. tuple1=("a","b") B. tuple1[2]=("a","b") C. tuple1=(5)*2 D. None of the above 2. Choose the correct option with respect to Python. A. Both tuples and...
-
Graphically show the optimal production level when the selling (market) price is between the AVC min and the AC min. Explain at which production levels the company has loss and where is the even...
-
A stock will pay no dividends for the next 9 years. Ten years from now, the stock is expected to pay its first dividend in the amount of $2.9. It is expected to pay a dividend of $3.2 exactly eleven...
-
Build a quantitative model to analyze the development and sale of a bicycle light. Assume that you could sell 20,000 units per year for five years at a sales price (wholesale) of $20 per unit and a...
-
1. For each of the following, briefly explain whether the situation violates any form of market efficiency, and why a. An announcement to issue equity results in a 3% decline in the stock price of...
-
Define a sequence of functions \(\left\{f_{n}(x)ight\}_{n=1}^{\infty}\) as \(f_{n}(x)=n^{2} x(1-x)^{n}\) for \(x \in \mathbb{R}\) and for all \(n \in \mathbb{N}\). a. Calculate \[f(x)=\lim _{n...
-
One hundred pounds of water at atmospheric pressure are heated from 60F to 200F. What is the enthalpy change? The internal energy change? Why is the difference between the internal energy change and...
-
A solution contains 22.4 g glucose (C 6 H 12 O 6 ) dissolved in 0.500 L of water. What is the molality of the solution? (Assume a density of 1.00 g/mL for water.) a) 0.238 m b) 44.8 m c) 0.249 m d)...
-
Which of the following compounds would give a positive Tollens test? (Remember that the Tollens test involves mild basic aqueous conditions.) (a) CH3CH2CH2COCH3 (b) CH3CH2CH2CH2CHO (c)...
-
Solving the following road-map problem depends on determining the structure of A, the key intermediate. Give structures for compounds A through K. hept-1-yne SOCI CH3CuLi (2) (CHj,S (2) Ho HCN (2)...
-
The UV spectrum of an unknown compound shows values of λmax at 225 nm (ε = 10,000) and at 318 nm 1e = 402. The mass spectrum shows a molecular ion at m/z 96 and a prominent...
-
Assume that a firm has weighted average cost of capital of 3.6% with a corporate tax rate of 21%. The pre-tax cost of debt is 3.1%. In case the firm would have no debt, the cost of equity for the...
-
Consider the stock of Aya Company, which will pay an annual dividend of $3 one year from today. The dividend will grow at a constant rate of 3%, forever.The market requires a 12% return on the...
-
Analyze the video presented on the link below. Also, please discuss or explain the video and relate it to personal finance. https://www.youtube.com/watch?v=nFY0HBkUm8o

Study smarter with the SolutionInn App


