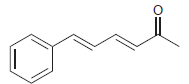
Use resonance structures to help you identify all sites of low electron density (δ+) in the following
Question:
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (17 reviews)
5...View the full answer

Answered By

OTIENO OBADO
I have a vast experience in teaching, mentoring and tutoring. I handle student concerns diligently and my academic background is undeniably aesthetic
4.30+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Use resonance structures to help you identify all sites of high electron density (δ-) in the following compound:
-
Phenol, C6H5OH, is a stronger acid then methanol, CH3OH, even though both contains an O ? H bond. Draw the structures of the anions resulting from loss of H+ from phenol and methanol, and use...
-
Addition of HC1 to 1-methoxycyclohexene yields 1-chloro-1-methoxycyclo- hexane as the sole product. Use resonance structures to explain why none of the other regioisomer isformed. HCI CI CI
-
Consider the cash flows in Table P6.7 for the following investment projects (MARR = 15%). Determine the annual equivalent worth for each project at i = 15% and determine the acceptability of each...
-
Video Concepts, Inc. (VCI) manufactures a line of DVD recorders (DVDs) that are distributed to large retailers. The line consists of three models of DVDs. The following data are available regarding...
-
x = 45, n = 150, 99% confidence Construct a confidence interval of the population proportion at the given level of confidence.
-
Draw a Graph for Income vs College! The New York Times created an interesting interactive graph on income versus percent of children who attend college. They ask you to first draw what you think the...
-
Salvania Corporation is considering investing in Farnorth Corporation, but is unsure about what level of ownership should be undertaken. Salvania and Farnorth have the following reported incomes:...
-
Assume the carrying capacity of the earth is 9 billion. Use the 1960s peak annual growth rate of 2.1% and population of 3 billion to predict the base growth rate and current growth rate with a...
-
It is often said that consumers receive free content online. Is this the case? Why or why not?
-
Suppose that you measured the product PV of 1 mol of a dilute gas and found that PV = 24.35 L atm at 0.00C and 33.54 L atm at 100.C. Assume that the ideal gas law is valid, with T = t(C) + a, and...
-
Liquid N 2 has a density of 875.4 kg m 3 at its normal boiling point. What volume does a balloon occupy at 298 K and a pressure of 1.00 atm if 3.10 10 3 L of liquid N 2 is injected into it? Assume...
-
There are very large numbers of charged particles in most objects. Why, then, dont most objects exhibit static electricity?
-
McCullough Hospital uses a job-order costing system to assign costs to its patients. Its direct materials include a variety of items such as pharmaceutical drugs, heart valves, artificial hips, and...
-
Jurvin Enterprises is a manufacturing company that had no beginning inventories. A subset of the transactions that it recorded during a recent month is shown below. a. $76,400 in raw materials were...
-
Record the July transactions. ( Credit account titles are automatically indented when amount is entered. Do not indent manually. List all debit entries before credit entries. If no entry is required,...
-
Compound and Continuous Compound Interest 1-Use compound interest formula (1) to find each or the indicated values. P=$5,000;i=0.005;n=36;A=?
-
How do emerging global challenges, such as climate change and invasive species, threaten the stability and functioning of ecosystems, and what adaptive strategies can be implemented to mitigate these...
-
Find the average rate of change of f from 0 to /2. f(x) = sin(x/2)
-
All of the following assets can be depreciated, except: (a) A bulldozer (b) A copper mine (c) A surgical robot (d) A conveyor belt
-
Synthesize the following compound starting with ethyne and 1-bromopentane as your only organic reagents (except for solvents) and using any needed inorganic compounds. Br Br
-
Predict features of their IR spectra that you could use to distinguish between the members of the following pairs of compounds. You may find the IR chart in the endpapers of the book and Table 2.1...
-
Deduce the structures of compounds A, B, and C, which all have the formula C6H10. As you read the information that follows, draw reaction flowcharts (roadmaps) like those in Problems 8.24 and 8.52....
-
1). The Electronic Corporation manufactures two electrical products: air conditioners and large fans. The assembly process for each is similar in that both require a certain amount of wiring and...
-
After reviewing oceanview marine company's case materials, and reviewing the unaudited account balances, what is your preliminary view on the client's ability to continue as a going concern? What is...
-
For each chemical reaction listed in the table below, decide whether the highlighted atom is being oxidized or reduced. reaction highlighted atom is being... neither oxidized reduced oxidized nor...

Study smarter with the SolutionInn App


