Hydrocarbon A, C 10 H 14 , has a UV absorption at ? max = 236 nm
Question:
Hydrocarbon A, C10H14, has a UV absorption at ? max = 236 nm and gives hydrocarbon B, C10H18, on catalytic hydrogenation. Ozonolysis of A followed by zinc/acetic acid treatment yields the following diketo dialdehyde:
(a) Propose two possible structures for A.
(b) Hydrocarbon A reacts with maleic anhydride to yield a Diels?Alder adduct. Which of your structures for A is correct?
(c) Write the reactions, showing starting material and products.
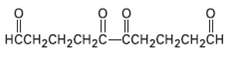
Transcribed Image Text:
ененонй оеко onрнм нссH-CH2CH2C —ҫсн-CH2CH2CH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (7 reviews)
a Hydrocarbon A must have two double bonds and two rings since the sole o...View the full answer

Answered By

Brown Arianne
Detail-oriented professional tutor with a solid 10 years of experience instilling confidence in high school and college students. Dedicated to empowering all students with constructive feedback and practical test-taking strategies. Effective educator and team player whether working in a school, university, or private provider setting. Active listener committed to helping students overcome academic challenges to reach personal goals.
4.60+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Propose two possible structures for a compound with molecular formula C 5 H 12 O that exhibits the following 13 C NMR and IR spectra: Carton 13 NMR 29.1 9.5- 73.8- 100 90 80 70 60 50 20 10 Chemical...
-
Propose two possible structures for a compound with molecular formula C 5 H 8 that produces an IR signal at 3300 cm -1 .
-
The following triene reacts with excess maleic anhydride to produce a compound with molecular formula C 14 H 12 O 6 . Draw the structure of this product (ignoring stereochemistry). Maleic anhydride...
-
Lamonda Corp. uses a job order cost system. On April 1, the accounts had the following balances: The following transactions occurred during April: (a) Purchased materials on account at a cost of...
-
How do you predict these pay rate changes at Parkland Health will affect its performance?
-
The 20-year A-rated bonds of Polly Cracker Company were initially issued at a 10 percent yield (paid semiannually). After 5 years, the bonds have been upgraded to AA. Such bonds are currently...
-
You are considering an investment in a tree farm. Trees grow each year by the following factors: The price of lumber follows a binomial lattice with \(u=1.20\) and \(d=9\). The interest rate is...
-
This exercise is an extension of Exercise 2-3B. Assume Jon Wallace completed the following additional transactions during March. Show the effect of each transaction on the basic elements of the...
-
Explain three (3) fundamentals of digital applications in a workplace What are the key digital applications that can be used in organisations? Explain three (3) key features of digital applications...
-
This assignment requires you to complete the 2022 tax reporting for a fictional woman named Anna Smith. Question 1 T1 - step 4 - line 66 This is Anna's taxable income Answer: Question 2 T1 - step...
-
Addition of HC1 to 1-methoxycyclohexene yields 1-chloro-1-methoxycyclo- hexane as the sole product. Use resonance structures to explain why none of the other regioisomer isformed. HCI CI CI
-
Adiponitrile, a starting material used in the manufacture of nylon, can be prepared in three steps from 1, 3-butadiene. How would you carry out thissynthesis? 3 steps H2C=CHCH=CH2 N=CCH2CH2CH2CH2C=N...
-
Return to the World Trade Organization webpage (http://stat.wto.org/StatisticalProgram/WSDBStatProgramHome.aspx?Language=E) and click "selection." For Subject, add "Merchandise trade indices" to the...
-
6. How much does architect Rivera receive in interest , if on April 10 they give him $20,312.20 for a capital that deposited in an account with interest of 14.4% on the previous August 23?
-
Normal America, Inc. (NA), has paid a year-end dividend every year for the past 10 years, as shown in the table below: Anne Share price as of December 31. Dividend of December 15. per share SP500...
-
In the market of Coke, supposed that its market-equilibrium price per can is $1.00 with 10,000 cans purchased (equilibrium), while the highest price a consumer is willing to pay is $3.00 per can. The...
-
A hedge fund wants to take advantage of current interest rates. It projects that the pound-to-dollar and euro-to-dollar exchange rates will remain the same one month from now in the worst-case...
-
A call option with 3 months remaining to maturity on an underlyingstock with no additional cash flows has an exercise price (X) of 50. Risk-free rate is 2%, and the stock is currently trading at...
-
Define a sequence of functions { f n ( x ) } n = 1 { f n ( x ) } n = 1 as f n ( x ) = n 2 x ( 1 x ) n f n ( x ) = n 2 x ( 1 x ) n for x [ 0 , 1 ] x [ 0 , 1 ] . Determine whether \[\lim _{n...
-
The age-old saying for investing is "buy low and sell high," but this is easier said than done. Investors who panic about falling prices sell their investments, which in turn lowers the price and...
-
What is the molarity of a 6.56% by mass glucose (C 6 H 12 O 6 ) solution? (The density of the solution is 1.03 g/mL.) SORT You are given the con- centration of a glucose solution in percent by mass...
-
(a) Simple aminoacetals hydrolyze quickly and easily in dilute acid. Propose a mechanism for hydrolysis of the following aminoacetal: (b) The nucleosides that make up DNA have heterocyclic rings...
-
The mass spectrum of unknown compound A shows a molecular ion at m/z 116 and prominent peaks at m/z 87 and m/z 101. Its UV spectrum shows no maximum above 200 nm. The IR and NMR spectra of A follow....
-
Show how you would accomplish the following synthetic conversions by adding an organolithium reagent to an acid. (a) (b) (c) Pentanoic acid heptan-3-one (d) Phenylacetic acid...
-
After the company issues an initial public offering and they desire to offer additional shares, one option is the seasoned equity offering. What are the differences between a convertible bond and a...
-
4. A stock provides a dividend yield of 5.0% paid semi- annually. The spot price of the stock is currently $500, and the risk-free rate is 7.5% with continuous compounding for all maturities. 1. What...
-
1. A 1000 bond issued on January 10, 2021 is paying coupons at a nominal interest rate of 7% payable semiannually. The bond is redeemed at par and matures on January 10, 2031. The nominal yield rate...

Study smarter with the SolutionInn App


