Question: All naturally occurring amino acids have the same relative configuration. All have the S absolute configuration, except for cysteine, which has the R configuration.Explain. CO.H
All naturally occurring amino acids have the same relative configuration. All have the S absolute configuration, except for cysteine, which has the R configuration.Explain.
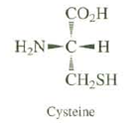
CO.H H,N-C-H CH,SH Cysteine
Step by Step Solution
3.36 Rating (168 Votes )
There are 3 Steps involved in it
In all of the amino acids except cysteine the CO 2 H group has priority ... View full answer

Get step-by-step solutions from verified subject matter experts
Document Format (1 attachment)
15-C-O-S (111).docx
120 KBs Word File


