An Add curved arrows to the following reactions to indicate the flow of electrons ineach: (a) H-CI
Question:
An Add curved arrows to the following reactions to indicate the flow of electrons ineach:
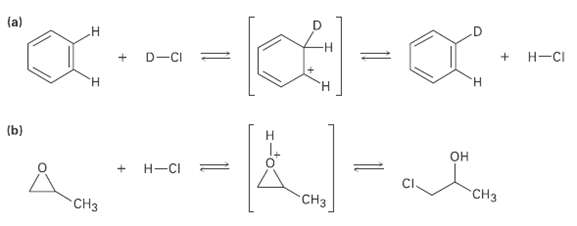
Transcribed Image Text:
(a) H-CI D-CI = H. H. (b) Н он Н-СI CHз CHз CHз
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (15 reviews)
b H 0...View the full answer

Answered By

Danish Sohail
My objective is to become most reliable expert for clients. For last 10 years I have been associated with the field of accounting and finance. My aim is to strive for best results and pay particular attention to client needs. I am always enthusiastic to help clients for issues and concerns related to business studies. I can work on analysis of the financial statements, calculate different ratios and analysis of ratios. I can critically evaluate stock prices based on the financial analysis and valuation for companies using financial statements of the business entity being valued with use of excel tools. I have expertise to provide effective and reliable help for projects in corporate finance, equity investments, financial accounting, cost accounting, financial planning, business plans, marketing plans, performance measurement, budgeting, economic research, risk assessment, risk management, derivatives, fixed income investments, taxation, auditing, and financial performance analysis.
4.80+
78+ Reviews
112+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Add curved arrows to the following reactions to indicate the flow of electrons for all of the bond-forming and bond-breaking steps. (a) (b) H.
-
Add curved arrows to the following polar reactions to indicate the flow of electrons in each: :Ci: (a) - :- + -N :CI: -NH (b) H CH: -Br: :Br: (c) :0: :CI: CI "
-
Add curved arrows to show how electrons must move to form the product from the reactants in the following equation, and locate any formal charges. :0 O: HO CH
-
One current practice is to calculate the years remaining for a depletable resource by taking the prevailing estimates of current reserves and dividing it by current annual consumption. How useful is...
-
Allen has purchased a whole life policy with a death benefit of $150,000. Assuming that he dies in 8 years and the average inflation has been 2%, what is the value of the purchasing power of the...
-
What is the free-rider problem, and how does it affect trade policy?
-
Derive Equations 5.26 and 5.30 . == Ef10f1Vf+ Em1m1Vm_ Ef1ff + Em1m1vm 1 Ef1Vf+ Em1Vm (5.26)
-
What occurs in each stage of the audit life cycle?
-
How does the principles, values, theories, techniques, methods and research approach to cognitive psychology school of thought match and attribute to general psychology?
-
A 5-year property costing $286,000 is placed in service at midyear in 2011. The property is depreciated using MACRS. Prepare a depreciation schedule for the property.
-
Despite the limitations of radical chlorination of alkanes, the reaction is still useful for synthesizing certain halogenated compounds. For which of the following compounds does radical chlorination...
-
A Follow the flow of electrons indicated by the curved arrows in each of the following reactions, and predicts the products thatresult: (b) :0- (a) -: "CH
-
Using online resources, find out when Viking was able to resume operations following the COVID-19 pandemic and how the health crisis affected Viking.
-
A. What is the linear programming model for problem below ? B. What is the complete optimal solution ? C. Will there be excess in any resources if so how much? 51=8 s2=0 53= 21) Biosphere...
-
Instructions on each Major Strategy/Assignment 1. Marketing Strategy Your marketing strategy consist of an evaluation of the Marketing Environment Forces (page 3 and 4 and Exhibit 1, page 12); b) an...
-
Profit center responsibility reporting for a service company Thomas Railroad Company organizes its three divisions, the North (N), South (S), and West (W) regions, as profit centers. The chief...
-
How much work is done lifting a 15 pound object from the ground to the top of a 25 foot building if the cable used weighs 1.5 pounds per foot? Question 2 A bucket begins weighing 20 pounds,...
-
Relevant Costs for Equipment Replacement Decision Health Scan, Inc. paid $50,000 for X-ray equipment four years ago. The equipment was expected to have a useful life of 10 years from the date of...
-
(a) Show that the relativistic Lagrangian \(L=-(1 / \gamma) m c^{2} e^{U / c^{2}}\) reduces to the Newtonian Lagrangian for a particle of mass \(m\) in a gravitational potential \(U\) in the slow...
-
1) The government decided to reduce taxes on fast-food to increase revenue. The government assumes that fast-food products have a) An inelastic demand b) An elastic demand c) A demand curve that is...
-
A conjugated hydrocarbon has an alternation of double and single bonds. Draw the molecular orbitals of the system of 1,3,5-hexatriene. If the energy required to excite an electron from the HOMO to...
-
(a) Give IUPAC substitutive names for the seven isomeric aldehydes and ketones with the formula C5H10O. (b) Give structures and names (common or IUPAC substitutive names) for all the aldehydes and...
-
Write a detailed mechanism for the formation of an acetal from benzaldehyde and methanol in the presence of an acid catalyst.
-
Outline all steps in the mechanism for the formation of a cyclic acetal from acetone and ethylene glycol (1,2-ethanediol) in the presence of gaseous HCl.
-
The Clifford Corporation has announced a rights offer to raise $36 million. The stock currently sells for $28 per share and there are 18 million shares outstanding. If the subscription price is set...
-
Superman Enterprises has just completed an initial public offering. The firm sold 4,700,000 new shares at an offer price of $17.00 per share. The underwritering spread was $0.78 a share. The firm...
-
ABV is a non-dividend paying stock whose current price is $50. Its volatility is 12%. Over each of the next two 6-month periods the stock price is expected to go up by 9% or down by 8%. The risk-free...

Study smarter with the SolutionInn App


