Despite the limitations of radical chlorination of alkanes, the reaction is still useful for synthesizing certain halogenated
Question:
Despite the limitations of radical chlorination of alkanes, the reaction is still useful for synthesizing certain halogenated compounds. For which of the following compounds does radical chlorination give a single monochloroproduct?
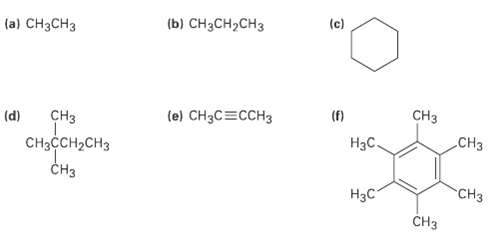
Transcribed Image Text:
(b) CH;CH,сHз (c) (a) CH3CH3 CHз (f) (e) CH3C=CCH3 CHз (d) CНз Нас. CHассH-CHз Cнз "CHз Нас CHз
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 81% (16 reviews)
The following compounds yield ...View the full answer

Answered By

S Mwaura
A quality-driven writer with special technical skills and vast experience in various disciplines. A plagiarism-free paper and impeccable quality content are what I deliver. Timely delivery and originality are guaranteed. Kindly allow me to do any work for you and I guarantee you an A-worthy paper.
4.80+
27+ Reviews
73+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Chlorination reactions of certain alkanes can be used for laboratory preparations. Examples are the preparation of chlorocyclopropane from cyclopropane and chlorocyclobutane from cyclobutane. What...
-
Which of the following halogenated compounds can be used successfully to prepare a Grignard reagent for alcohol synthesis by subsequent reaction with an aldehyde or ketone? Which ones cannot and why?...
-
Radical chlorination of alkanes is not generally useful because mixtures of products often result when more than one kind of CH bond is present in the substrate. Draw and name all monochloro...
-
To anticipate subsequent chapters where more complicated renewable resource models are introduced, consider a slight modification of the two-period depletable resource model. Suppose a biological...
-
Suppose you are 45 and have a $10,000 face amount, 15-year, limited-payment, participating policy (dividends will be used to build up the cash value of the policy). Your annual premium is $350. The...
-
Suppose that Canada produces 1.0 million bicycles a year and imports another 0.4 million; there is no tariff or other import barrier. Bicycles sell for $400 each. * Parliament is considering a $40...
-
The interfacial shear stresses, \(\tau\), and the fiber normal stresses, \(\sigma_{f}\) acting on a differential element at a distance \(x\) from the end of the fiber are shown in Figure 6.4, where...
-
John conducted a repeated-measures study to measure self-esteem among victims of domestic violence before and after they complete an 8-week program for abuse victims with 10 individuals. He...
-
. What is a non-political socially-appropriate response to someone who is exhibiting prejudice? What are the responsibilities of a social psychology researcher when the researcher encounters...
-
Selecting the Best Two to Four Warehouse Locations for Als AthleticsMini Case Study. This hands-on example will give you your first opportunity to further your understanding of the books concepts by...
-
Radical chlorination of pentane is a poor way to prepare 1-chioropentane, but radical chlorination of neopentane, (CH3)4C, is a good way to prepare neopentyl chloride, (CH3)3CCH2C1. Explain.
-
An Add curved arrows to the following reactions to indicate the flow of electrons ineach: (a) H-CI D-CI = H. H. (b) -I CH CH CH
-
Calculating the standard error of the mean Given s = 7.50 and N = 225, calculate s M .
-
After reading the article: Work-Life Balance Issues: Changing When and How Work Gets Done , respond to the following: It is mentioned in the reading that even when organizations offer work-life...
-
The draft statement of profit or loss for Harris Ltd for the 9 months ending 30 September 2022 is given below: Sales Cost of sales Gross profit Administration salaries Insurance Rent Advertising...
-
Describe a time when you, or someone you know, received an annual evaluation appraisal. How did the supervisor approach the meeting? Did you feel it was a fair assessment of your performance? Did you...
-
7 Skipped eBook References b. Prepare journal entries (including any adjusting entry as of the end of the month) to record these items using the procedure of initially crediting the Revenue account...
-
2. Write a program that examines a series of card hands and stores the ones whose totals are
-
The Lagrangian of a particle of mass \(m\) and charge \(e\) in a uniform, static magnetic field \(\mathbf{B}\) is given by \(L(\mathbf{x}, \dot{\mathbf{x}})=\frac{1}{2} m \dot{\mathbf{x}}^{2}+(e / 2...
-
Cobb Manufacturing Company uses a process cost system and average costing. The following production data is for the month of June 2011. Production Costs Work in process, beginning of the month:...
-
An elusive intermediate of atmospheric reactions of HONO may be nitrosyl O-hydroxide, HOON. Electronic structure calculations seem to indicate that HOON is best represented by a combination of...
-
Predict the major products formed when: (a) Toluene is sulfonated. (b) Benzoic acid is nitrated. (c) Nitrobenzene is brominated. (d) Isopropylbenzene reacts with acetyl chloride and AlCl3. If the...
-
Use resonance theory to explain why the hydroxyl group of phenol is an activating group and an ortho-para director. Illustrate your explanation by showing the arenium ions formed when phenol reacts...
-
Phenol reacts with acetic anhydride in the presence of sodium acetate to produce the ester phenyl acetate: The CH3COOi- group of phenyl acetate, like the -OH group of phenol (Practice Problem 15.8),...
-
On a particular day, there were 300 stocks that advanced on the NYSE and 800 that declined. The volume in advancing issues was 1000 and the volume in declining issues was 3000. What is the trin ratio?
-
A coupon bond that pays interest of $30 annually has a par value of $1000, matures in 6 years, and is selling today at $850. What is the yield to maturity on this bond?
-
A callable bond pays annual interest of $40, has a par value of $1000, matures in 4 years but is callable in 3 years at a price of $1100, and has a value today of $1020. What is the yield to call on...

Study smarter with the SolutionInn App


