An aqueous glycerol solution weighing 100.0 mg was treated with 50.0 mL of 0.083 7 M Ce
Question:
An aqueous glycerol solution weighing 100.0 mg was treated with 50.0 mL of 0.083 7 M Ce4+ in 4 M HClO4 at 60°C for 15 min to oxidize the glycerol to formic acid:
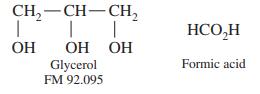
The excess Ce4+ required 12.11 mL of 0.044 8 M Fe2+ to reach a ferroin end point. What is the weight percent of glycerol in the unknown?
Transcribed Image Text:
CH, — CH— СH, | HCO,H OH ОН ОН Formic acid Glycerol FM 92.095
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (10 reviews)
One mole of glycerol requires eight moles of Ce 4 500 mL of 0083 7 M Ce 4 418...View the full answer

Answered By

AJIN KURIAKOSE
I HAVE ELECTRONICS ENGINEERING DEGREE..AND MY AREA OF INTEREST IS MATHEMATICS,CONTROL SYSTEM,NETWORK,DIGITAL
4.70+
21+ Reviews
32+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Chemical Engineering questions
-
Nitrite (NO 2 - ) can be determined by oxidation with excess Ce 4+ , followed by back titration of the unreacted Ce 4+ . A 4.030-g sample of solid containing only NaNO 2 (FM 68.995) and NaNO 3 was...
-
Aqueous solutions of the amino acid L-isoleucine (Ile) are prepared by putting 100.0 grams of pure water into each of six flasks and adding different precisely weighed quantities of Ile to each...
-
Penicillin is produced by fermentation and recovered from the resulting aqueous broth by extraction with butyl acetate. The penicillin distribution coefficient K (mass fraction of penicillin in the...
-
Explain how the structure of the cash flow statement represents another subsection of the accounting equation.
-
Peralta Company borrows $60,000 on July 1 from the bank by signing a $60,000, 10%, one-year note payable. (a) Prepare the journal entry to record the proceeds of the note. (b) Prepare the journal...
-
1. A new model of golf shoes is listed at RM110 per pair. During the sale, a trade discount of 14% was offered. a) What is the amount of the trade discount offered per pair of shoes? b) What is the...
-
Fill in the Blank. Univariate distributions describe the probability distributions of __________ random variables.
-
a. Develop an AON network for this problem. b. Create a spreadsheet to calculate and summarize the earliest and latest start and finish times, the slack for each activity, and the critical...
-
Explore key security features implemented at the kernel level, such as mandatory access controls (e.g., SELinux, AppArmor) and address space layout randomization (ASLR). How do these features...
-
Consider a two-stage cascade refrigeration system operating between -80oC and 80oC. Each stage operates on an ideal vapor-compression refrigeration cycle. Upper cycle use R-12 as working fluid, lower...
-
A 50.00-mL sample containing La 3+ was treated with sodium oxalate to precipitate La 2 (C 2 O 4 ) 3 , which was washed, dissolved in acid, and titrated with 18.04 mL of 0.006 363 M KMnO 4 . Calculate...
-
Consider the titration of 100.0 mL of 0.010 0 M Ce 4+ in 1 M HClO 4 by 0.040 0 M Cu+ to give Ce 3+ and Cu 2+ , using Pt and saturated Ag | AgCl electrodes to find the end point. (a) Write a balanced...
-
Calculate the change in availability (kW) of the two flows in Problem 9.61.
-
The cylinder below has a mass of 500 kg. All surfaces are smooth. A, B and C are frictionless Pins. The members AB and BC are considered to have a negligible weight. Calculate the following: a)...
-
Compute the horizontal force P required to prevent the 900 lb block, shown below from sliding down the plane. The coefficient of friction between the block and the plane is 0.20. W = 900 lb 35 ASSI 35
-
The ladder shown is supported by a horizontal floor and a vertical wall. It is 12 ft long, weighs 30 lb (assumed to be concentrated at its mid-length), and supports a person weighing 175 lb at point...
-
5-3: Consider a natural gas mixture with mass fractions of methane and ethane of 0.75 and 0.25, respectively. Suppose that a large reservoir contains 106 ft of this natural gas at conditions of 2000...
-
As a senior manufacturing engineer at a company that makes welded tool cabinets and related products, you are asked to optimize a dissimilar metal beam welding process that exists as the critical...
-
In 2024, Kadlec Company had the following transactions related to the purchase of a property. All transactions were for cash unless otherwise stated. Jan. 12 Purchased real estate for a future plant...
-
a. Show that the expansion of q(x) in ascending powers of x can be approximated to 10 2x + Bx 2 + Cx 3 where B and C are constants to be found. b. Find the percentage error made in using the series...
-
Why is it advantageous to use large particles (50 m) for solidphase extraction, but small particles (5 m) for chromatography?
-
In 2002, workers at the Swedish National Food Administration discovered that heated, carbohydrate-rich foods, such as french fries, potato chips, and bread, contain alarming levels (0.1 to 4 g/g) of...
-
Many metals in seawater can be preconcentrated for analysis by coprecipitation with Ga(OH)3. A 200 -L HCl Filtered Unfiltered Fe 1000 1 200 Depth (m)
-
Design a Turing Machine (TM) for the following language, give formal description of the TM and draw state diagram: {w|w is a string over the alphabet {0, 1} and number of 1's in w is divisible by 3}....
-
Jimmy John Hunter owns a small farm outside Winona and was getting very frustrated with turkey vultures that pester his sleeping livestock to the point where his chickens are laying fewer eggs. A few...
-
If the beginning cash balance is $ 5,000, the net cash inflow from operating activities is $20.000, net cash outflow from investing activities is (15,000) and net inflows from financing activities is...

Study smarter with the SolutionInn App


