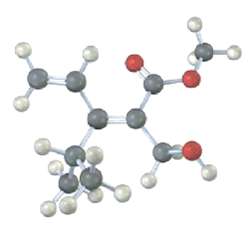
Question: Assign stereochemistry (E or Z) to the double bond in the following compound, and convert the drawing into a skeletal structure (red0):
Assign stereochemistry (E or Z) to the double bond in the following compound, and convert the drawing into a skeletal structure (red0):
Step by Step Solution
3.25 Rating (157 Votes )
There are 3 Steps involved in it
High L... View full answer

Get step-by-step solutions from verified subject matter experts
Document Format (1 attachment)
22-C-OC-A (89).docx
120 KBs Word File


