Name the following alkenes, and tell which compound in each pair is more stable CH {a) H2C=CHCH2CH3
Question:
Name the following alkenes, and tell which compound in each pair is more stable
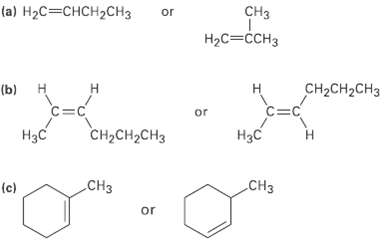
Transcribed Image Text:
CHз {a) H2C=CHCH2CH3 or Нас—ссHз CH2CH2CH3 Н (b) Н C=C or H3C CH-сH,CHз Нас CНз CHз (c) or
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 63% (11 reviews)
a b c More stable H 1 CC H CH3 2Methylpropene disubstituted double bond H C...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Which species in each pair is a better oxidizing agent under standard-state conditions? (a) Br2 or Au3+. (b) H2 or Ag+. (c) Cd2+ or Cr3+. (d) O2 in acidic media or O2 in basic media.
-
Which species in each pair is a better reducing agent under standard-state conditions? (a) Na or Li. (b) H2 or I2. (c) Fe2+ or Ag. (d) Br- or Co2+.
-
1. Which compound in each pair has a higher melting or boiling point? Explain. a. MgO or NaCI (melt) b. HBr or Kr (boil) c. CaBr, or CaCli(melt) d. SiH, or PH, (boil) 2. The molar heat of...
-
(a) By what percentage does your rest mass increase when you climb 30 m to the top of a ten-story building? Are you aware of this increase? Explain. (b) By how many grams does the mass of a 120-g...
-
In what way do effective teachers practice contingency leadership?
-
Exchange rates can experience sudden changes as well as long-run patterns. a. Plotthe daily U.S. dollar-British pound exchange rate (FRED code: DEXUSUK) for the first half of 2016 and identify the...
-
INTEL: Building Blocks Inside Out Visit www.pearsonglobaleditions.com/malhotra to read the video case and view the accompanying video. Intel: Building Blocks Inside Out demonstrates the critical role...
-
Using Figure 7-5 as an example, redraw Figure 7-13 using an enterprise information system that processes a shared database. Explain the advantages of this system over the paper-based system in Figure...
-
EXERCISE Select a country as a target market and fill in the following chart based on the potential barriers to entry for this target market. Explain the rationale behind the strategies chosen to...
-
1: Using variable elimination (by hand!), compute the probability that a student who did well on the test actually understood the material, that is, compute P(+u| + e). 2: For the above Bayesian...
-
Assign stereochemistry (E or Z) to the double bond in the following compound, and convert the drawing into a skeletal structure (red0):
-
Predict the products of the following reactions: (b) CH CHCCHH2CH (a) HCI H (d) CH CH2 (c) HBr CH-CH3CH2 H20 H2SO, (Addition of H20 occurs.)
-
Why are accurate fraud statistics hard to find?
-
Calculate the overall on-prem cost for the e-commerce architecture. State all the assumptions. You can omit the costs related to the IT staff
-
Discuss the challenges and methods involved in calculating "Consolidated Earnings per Share (EPS)". How do intercompany transactions and noncontrolling interests impact the EPS calculation?
-
Explore the intricacies of "Taxation of Consolidated Companies". How do tax regulations and implications differ for consolidated entities compared to standalone companies? How do intercompany...
-
Describe the importance of presenting a comprehensive and accurate Consolidated EPS. How do factors like stock splits, dividends, and changes in ownership interest affect its calculation?
-
A gas pump measures volume of gas to the nearest 0.07 gallon. Which measurement shows an appropriate level of precision for the pump?
-
Draw a Nyquist diagram for each of the following systems and compare the result with that obtained using the MATLAB command Nyquist. (a) \(K G(s)=\frac{K(s+2)}{s+10}\) (b) \(\quad K...
-
Reichenbach Co., organized in 2018, has set up a single account for all intangible assets. The following summary discloses the debit entries that have been recorded during 2018 and 2019. Instructions...
-
How much energy is released when 65.8 g of water freezes?
-
Draw the products of each of the following reactions indicated by the curved-arrow notation. (a) (b) CH CH NH2
-
Use the curved-arrow notation to indicate the flow of electrons in each of the transformations given in Fig. P3.33. (a) (b) (c) (d) (CHJ.NH-CH.-CH-C-OC-H5 BrC2 HC CH CH3 CH2 CH CH C: T:0:
-
Predict the products of each of the following reactions, and explain your reasoning. Use the curved-arrow notation to help you, and show the notation. (a) (b) AICI3 011 + H,N-CH-CH, -
-
Techuxia Corporation worked on four jobs during October: Job A256, Job A257, Job A258, and Job A260. At the end of October, the job cost sheets for these jobs contained the following data: Beginning...
-
You are considering your retirement plans. You would like to buy a NICE RV and see the country. This will cost $281,000 You have $37,000 to open the account and you will deposit $685 at the end of...
-
Find functional dependencies. Date Slot StartTime End Time Class Subject 21/10/2018 1 7:00 8:30 SE1023 Database 21/10/2018 2 8:45 10:15 SE1022 Database 21/10/2018 3 10:30 12:00 SE1016 Database Date...

Study smarter with the SolutionInn App


