For each molecular geometry, list the number of total electron groups, the number of bonding groups, and
Question:
For each molecular geometry, list the number of total electron groups, the number of bonding groups, and the number of lone pairs on the central atom.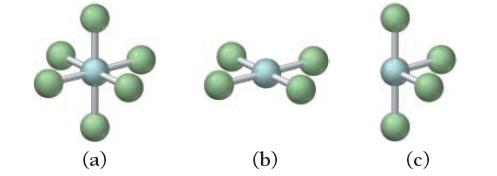
Transcribed Image Text:
နာ (a) (b) (c)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
a Total electron groups 4 Bonding groups 4 Lone pairs 0 ...View the full answer

Answered By

Muqadas Javed
I am a mentor by profession since seven years. I have been teaching on online forums and in universities. Teaching is my passion therefore i always try to find simple solution for complicated problems or task grasp them so that students can easily grasp them.I will provide you very detailed and self explanatory answers and that will help you to get good grade. I have two slogans: quality solution and on time delivery.
4.60+
24+ Reviews
144+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
For each molecular geometry, list the number of total electron groups, the number of bonding groups, and the number of lone pairs on the central atom. (a) (b) (c)
-
Describe a hybridization scheme for the central S atom in the molecule SF 4 that is consistent with the geometric shape pictured in Table 10.1. Which orbitals of the S atom are involved in overlaps,...
-
Describe a hybridization scheme for the central Cl atom in the molecule ClF 3 that is consistent with the geometric shape pictured in Table 10.1. Which orbitals of the Cl atom are involved in...
-
finish this The five components, which are scope, references, configuration item, baseline, and verification, are vital to ensure the configuration management plan establishes control, ensures...
-
All future costs are relevant in decision making. Do you agree? Why?
-
In the early 1990s, the amount of time that elapsed between purchasing a stock and actually obtaining that stock was five business days. This period was known as the settlement period. The settlement...
-
For the given table of observed frequencies: a. Compute the row totals, the column totals, and the grand total. b. Construct the corresponding table of expected frequencies. c. Compute the value of...
-
Outrageous Bubbles, Inc., produces multicolored bubble solution used for weddings and other events. The company's master budget income statement for March follows. It is based on expected sales...
-
Question Two: For the following stable growth firm , calculate: Cost of equity using CAPM, having risk free rate to be 2%, market risk premium 8%, and equity beta 1.4 Current growth rate with the...
-
Determine the electron geometry, molecular geometry, and idealized bond angles for each molecule. In which cases do you expect deviations from the idealized bond angle? a. PF 3 b. SBr 2 c. CHCl 3 ...
-
A molecule with the formula AB 3 has a trigonal planar geometry. How many electron groups are on the central atom?
-
Economist Robert Gordon has written the following: During 1939, more than any other year in the dismal Depression decade, the American economy exhibited every evidence of slipping into a...
-
Suppose that $24,000 is invested at a rate of 5% interest compounded monthly. Determine how much the investment is worth after 10 years.
-
A line passes through the point (3,9), and it's parallel to the line x = -9. Find an equation for this line. An equation for this line is
-
China's military doctrine closely associates cyber-attacks and nuclear high-altitude electromagnetic pulse (HEMP) attacks, as part of a combined operation in what they call Total Information Warfare....
-
The following production data were taken from the records of the finishing department for June: Line Item Description Amount Inventory in process, June 1 , 2 5 % completed 3 8 0 units Transferred to...
-
Power can affect the workplace in both positive and negative ways. Describe one element of power that has the potential to be positive and explain your reasoning. Please provide the reference you...
-
What are sources of risk facing a firm which are reflected on its income statement?
-
List four items of financial information you consider to be important to a manager of a business that has been operating for a year.
-
The member is to be made from a steel plate that is 0.25 in. thick. If a 1-in. hole is drilled through its center, determine the approximate width w of the plate so that it can support an axial force...
-
Determine the maximum normal stress developed in the bar when it is subjected to a tension of P = 2 kip. 0.125 in. 1.875 in. 1.25 in. r = 0.25 in. 0.75 in.
-
Determine the maximum axial force P that can be applied to the bar. The bar is made from steel and has an allowable stress of Ï allow = 21 ksi. 0.125 in. 1.875 in. 1.25 in. r = 0.25 in. 0.75 in.
-
On June 30, 2020, Wisconsin, Inc., issued $274,800 in debt and 17,700 new shares of its $10 par value stock to Badger Company owners in exchange for all of the outstanding shares of that company....
-
12 If tant= a and
-
The Monty Theater opened on April 1. All facilities were completed on March 31. At this time, the ledger showed No. 101 Cash $7,740, No. 140 Land $10,260, No. 145 Buildings (concession stand,...

Study smarter with the SolutionInn App


