Calculate product rates and compositions, stage temperatures, and interstage vapor and liquid flow rates and compositions for
Question:
Calculate product rates and compositions, stage temperatures, and interstage vapor and liquid flow rates and compositions for an absorber having four equilibrium stages with the specifications in figure. Assume the oil isnC10.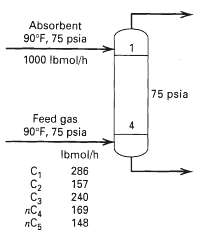
Transcribed Image Text:
Absorbent 90°F, 75 psia 1000 Ibmol/h 75 psia Feed gas 90°F, 75 psia 4 Ibmol/h 286 C2 C3 пС 157 240 169 148 తొలోలొలె
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 54% (11 reviews)
As shown below when the gas feed was flashed it was found to contain 72 mol liquid The c...View the full answer

Answered By

Diksha Bhasin
I have been taking online teaching classes from past 5 years, i.e.2013-2019 for students from classes 1st-10th. I also take online and home tuitions for classes 11th and 12th for subjects – Business Studies and Economics from past 3 years, i.e. from 2016-2019. I am eligible for tutoring Commerce graduates and post graduates. I am a responsible for staying in contact with my students and maintaining a high passing rate.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Chemical Engineering questions
-
Calculate product compositions, stage temperatures interstage vapor and liquid flow rates and compositions, reboiler duty, and condenser duty for the following distillation-column specifications Feed...
-
Calculate the equilibrium constant for the reaction O2 2O at temperatures of 537 R and 10 000 R.
-
Calculate the IRR of each of the four stages of the cost reduction proposal in Problem 2 of Exercise 16.2. Based on the IRR investment criterion, which stages should be approved at a 14% cost of...
-
In July 2009, Malcolm bought a piece of land for 40,000. In June 2016 he sold part of the land for 11,000. This disposal was not caused by a compulsory purchase and was his only disposal of land in...
-
The following information is for Kon Inc. for the year ended December 31, 2012: Common shares, Jan. 1..................................$20,000 Common shares issued during year..............10,000...
-
A 2-kg sphere S is being moved in a vertical plane by a robotic arm. When the angle is 30, the angular velocity of the arm about a horizontal axis through O is 50 deg /s clockwise and its angular...
-
With reference to Exercise 3.1, suppose that the points \((0,0),(0,1),(0,2),(0,3),(1,0),(1,1),(1,2),(1,3)\), \((2,0),(2,1),(2,2),(2,3),(3,0),(3,1),(3,2)\), and \((3,3)\) have the probabilities...
-
Does competition help or hurt the valuation of a business? Explain.
-
Sandra thinks that people who have lots of friends tend to have higher happiness ratings. To see if she is correct, Sandra recruits a sample of 10 people and asks them each how many friends they...
-
Golf Corp. (GC), a calendar-year accrual-method corporation, held its directors meeting on December 15 of year 1. During the meeting the board of directors authorized GC to pay a $75,000 charitable...
-
An absorber is to be designed for a pressure of 75 psia to handle 2,000 lbmol/h of gas at 60?F having the following composition.The absorbent is an oil, which can be treated as a pure component...
-
In Example 10.4, temperatures of the gas and oil, as they pass through the absorber, increase substantially. This limits the extent of absorption. Repeat the calculations with a heat exchanger that...
-
How high can the minimum wage go before it has a negative effect on employment and why? Minimum wage has two different results in perfectly competitive and classical monophony. In case of perfectly...
-
Discuss decision-making as a structure using the Aldi versus Walmart example. a. What kind of decisions need to be made to change behaviors with clients? b. What kind of decisions need to be made to...
-
We assume that z 0 for all t (i.e. the boat never changes tack). We want to determine the fastest trajectory z(x). Write the expression of the time dt to go, on this trajectory, from x to x + dx in...
-
Explain the result physically. A skier slides down a snowy plane slope. The plane makes an angle with respect to the horizontal direction. The skier is in the vertical field of gravity, of...
-
Using the observed value of molecular diffusion of heat in water, estimate how long it would take for a temperature anomaly to mix from the top of the ocean to the bottom, assuming that molecular...
-
In what ways do you see people buying into excuses or powerlessness?
-
A publicly traded company is any business entity _______________. a. That is traded on a domestic stock exchange or over-the-counter market at the local, regional, or national level b. That is...
-
1. True or False. Pitfalls to consider in a statistical test include nonrandom samples, small sample size, and lack of causal links. 2. Because 25 percent of the students in my morning statistics...
-
Identify three self-regulating agencies and explain which industries they regulate. Speculate as to why an industry would self- regulate.
-
Derive (15-44) and use it to solve the following problem. Sulfate ion is to be removed from 60 L of water by exchanging it with chloride ion on 1 L of a strong-base resin with relative molar...
-
For mass transfer outside a single spherical particle that is not close to a wall or other particles, what is the smallest value of the Sherwood number? What is the basis for this value?
-
Within a porous particle, why are mass and heat transfer not analogous?
-
If you invest $250 a month for 40 years at 10% interest. How much money will you have.
-
Watch the Video "Types of Risks - Risk Management in Banks" https://youtu.be/8QjCdwhPT8U Provide a one paragraph answer to each of the following questions: In your opinion, what type of bank risk has...
-
4. A product manager prepared the following forecasts for a product line: 0 1 2 3 4 5 6 and later + + + + + $2m $2m $3m $4m $4m g=2.5% If the appropriate discount rate is 8.5%, what is the present...

Study smarter with the SolutionInn App


