Calculate the pKa for these compounds. a) HCOH (K 1.75 X 104) b) CHCH3 (K = 10-50)
Question:
Calculate the pKa for these compounds.
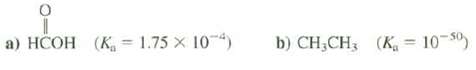
Transcribed Image Text:
a) HCOH (K 1.75 X 104) b) CH₂CH3 (K = 10-50)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (10 reviews)
p K a...View the full answer

Answered By

Ali Khawaja
my expertise are as follows: financial accounting : - journal entries - financial statements including balance sheet, profit & loss account, cash flow statement & statement of changes in equity -consolidated statement of financial position. -ratio analysis -depreciation methods -accounting concepts -understanding and application of all international financial reporting standards (ifrs) -international accounting standards (ias) -etc business analysis : -business strategy -strategic choices -business processes -e-business -e-marketing -project management -finance -hrm financial management : -project appraisal -capital budgeting -net present value (npv) -internal rate of return (irr) -net present value(npv) -payback period -strategic position -strategic choices -information technology -project management -finance -human resource management auditing: -internal audit -external audit -substantive procedures -analytic procedures -designing and assessment of internal controls -developing the flow charts & data flow diagrams -audit reports -engagement letter -materiality economics: -micro -macro -game theory -econometric -mathematical application in economics -empirical macroeconomics -international trade -international political economy -monetary theory and policy -public economics ,business law, and all regarding commerce
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Using the pKa values of analogous compounds in Table 3.1, predict which would be the stronger base. (a) (b) (c) (d) or (CHa),Cor O: HO or
-
Compounds like CCl2F2 are known as chlorofluorocarbons, or CFCs. These compounds were once widely used as refrigerants but are now being replaced by compounds that are believed to be less harmful to...
-
The pKa of protonated acetone is about -7.5 and the pKa of protonated hydroxylamine is 6.0. a. In its reaction with hydroxylamine at pH = 4.5 (Figure 18.2), what fraction of acetone will be present...
-
Q1-Mutual funds provide the following for their shareholders. A. diversification B. professional management C. record keeping and administration D. all of these options
-
Office Magic Ltd manufactures ergonomic office equipment. The standard cost for material and labour is $190 per unit. This includes 8 kilograms of direct material at a standard cost of $5 per...
-
Refer to Exercise 16.11. a. Test to determine whether there is evidence of a linear relationship between distance to the nearest fire station and percentage of damage. b. Estimate the slope...
-
For a sample of size n = 20, the following values were obtained: b0 = 1.05, b1 = 4.50, se = 0.54, (x x )2 = 10.9, x = 8.52. Construct a 95% confidence interval for the mean response when x = 10.
-
Beta Corporation is in the process of completing a contract to produce 150 units for a given customer. The contract consisted of R&D, testing and qualification, and full production. The industrial...
-
The following exercise may be answered manually or with the assistance of a computer. Assume that the random variable is normally distributed. You may need to use the appropriate appendix table to...
-
Maria Chavez owns a catering company that serves food and beverages at parties and business functions. Chavezs business is seasonal, with a heavy schedule during the summer months and holidays and a...
-
Which of these species can behave as a Lewis base? a) CH0CH,CH, CH3 d) CH3NH + b) CH3CHCH3 0: I.. e) CHCOH c) CH3NH
-
Calculate the Ka for these compounds. a) HC=CH (pK = 25) b) HC N (pK = 9.31)
-
During its first year of operations, Cupola Fan Corporation issued 30,000 of $1 par Class B shares for $385,000 on June 30, 2018. Share issue costs were $1,500. One year from the issue date (July 1,...
-
The following sum 1 5 + 1+ 5 n 1 n is a right Riemann sum for a certain definite integral 1 + 10 n 5 n + 1+ 1 15 n 52 L f(x) dx using a partition of the interval [1,6] into n subintervals of equal...
-
Which do you think are more important to emphasize in a literature review, the opinions of experts in the field or related studies and why? Describe the kind of behaviours that individual team...
-
Channel Tunnel Inc. plans to build an additional 23-mile-long tunnel under the English Channel for added train service. The cost (NINV) of the tunnel is expected to be $2.4 billion. Net cash inflows...
-
Suppose that the Forward price of the ABC stock is 100. A call option on ABC with exercise price 100 is trading at $24 and a put option with the same exercise price is trading at $18. All are with...
-
Develop a Secure Java EE "Product Catalog Management Application" for the functionality described in the requirements section. For each product in the catalog, product data should include product...
-
Explain the concept of depreciation as a noncash expense. Does this mean it has no impact at all on a ventures cash flows?
-
a. What is the cost of borrowing if Amarjit borrows $28 500 and repays it over a four-year period? b. How many shares of each stock would he get if he used the $28 500 and invested equally in all...
-
Prove the identity. sin(/2 + x) = cos x
-
Show how the Wittig reaction might be used to prepare the following alkenes. Identify the alkyl halide and the carbonyl components that would he used. (b) (a)
-
How would you use a Grignard reaction on an aldehyde or ketone to synthesize the following compounds? (a) 2-Pentanol (b) 1-Butanol (c) 1-Phenylcyclohexanol (d) Diphenyl methanol
-
Aldehydes can be prepared by the Wittig reaction using (methoxymethylene)-triphenylphosphorane as the Wittig reagent and then hydrolyzing the product with acid. For example, (a) How would you prepare...
-
what are your thoughts on the answers below about Effective listeners After selecting "Dating Violence" in the difficult tab of the general listening quiz section, I scored a 5/5. I found it quite...
-
Review this between the U.S. Deputy Secretary of Defense and the U.S. Secretary of the Air Force. This is an historic exchange that introduces the first attempt by the military to address sexual...
-
You have decided to purchase a computer. Research some options of a computer you wish to buy on the Internet.The bank has given you some different options on loans that you would like to look at....

Study smarter with the SolutionInn App


