Aldehydes can be prepared by the Wittig reaction using (methoxymethylene)-triphenylphosphorane as the Wittig reagent and then hydrolyzing
Question:
Aldehydes can be prepared by the Wittig reaction using (methoxymethylene)-triphenylphosphorane as the Wittig reagent and then hydrolyzing the product with acid. For example,
(a) How would you prepare the necessary phosphorane?
(b) Propose a mechanism for the hydrolysisstep.
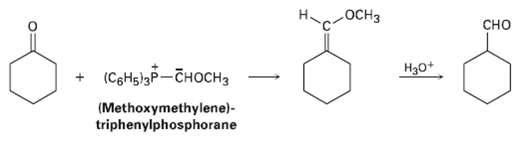
Transcribed Image Text:
OCH3 сно (СоНiзр— сноснз H30+ (Methoxymethylene)- triphenylphosphorane
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 72% (11 reviews)
a b Ph3P BrCHOCH3 HOCH3 H0H protonation ...View the full answer

Answered By

Sinmon Warui Kamau
After moving up and down looking for a job, a friend introduced me to freelance writing. I started with content writing and later navigated to academic writing. I love writing because apart from making a living out of it, it is also a method of learning and helping others to learn.
5.00+
40+ Reviews
45+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
How would you prepare 72.5 g of an aqueous solution that is 5.00% potassium iodide, KI, by mass?
-
How would you prepare 455 g of an aqueous solution that is 6.50% sodium sulfate, Na2SO4, by mass?
-
How would you prepare 2.00 L of (a) 0.10 M KOH from the solid? (b) 0.010 M Ba(OH)2 8H2O from the solid? (c) 0.150 M HCl from a reagent that has a density of 1.0579 g/mL and is 11.50% HCl (w/w)?
-
Every human being fears to fail. This is the same case with the leaders. Most of the leaders are afraid of some things, but failing is the most feared among all of them. The loss of direction among...
-
Various conflict management styles are depicted on PPT 9-7. For each of the participants in the scenario, describe which style(s) they used and cite examples to support your answer.
-
What value of A makes the two annual cash flows equivalent at 13% interest compounded annually? $120 $120 $120 $100 $100 Years Years
-
Assuming factorability of the complex degree of coherence of polarized thermal light, demonstrate that when the photodetector area is much larger than the coherence area of the incident light, the...
-
A 30.0-liter cylinder of a gas containing 97.0 mole% CO and 3.0% CO2 is delivered to your plant. You sign the receipt for it, noting that the gauge on the tank reads 2000 psi. Several days later you...
-
A wire is first bent into the shape of a square. Each side of the square is 10 cm long. Then the wire is unbent and reshaped into a rectangle. If the length of the rectangle is 13 cm, what is its...
-
Wok Yow Imports, Inc., is a rapidly growing, closely held corporation that imports and sells Asian style furniture and accessories at several retail outlets. The equity owners are considering selling...
-
How would you use a Grignard reaction on an aldehyde or ketone to synthesize the following compounds? (a) 2-Pentanol (b) 1-Butanol (c) 1-Phenylcyclohexanol (d) Diphenyl methanol
-
When 4-hydroxyhutanal is treated with methanol in the presence of an acid catalyst, 2-mcthoxytctrahydrofuran is formed. Explain. -H C HCH2H2CH C
-
The following items were taken from the financial statements of J. Kung Enterprises. (All amounts are in thousands.) Instructions Prepare a classified statement of financial position in good form as...
-
Suppose rogers bond f=1000 , cr= 5.34 maturity= March 1 2027. interest /coupon payment dates are March 1 & September 1 Determine accrued interest of Tuesday jan 18 2021:
-
Sea Grove Beach Corporation is executing an IPO with the following characteristics. The company will sell 12 million shares at an offer price of $20 per share and the investment bank underwriting the...
-
Why is forecasting factor premiums necessary? Why doesnt this same logic apply to factor exposures?
-
(3) (8 pts) Using the explicit function f(x) given, write out (but do NOT calculate) the complete right Riemann sum R4 for f(x) = 1 +2|x on the interval [-2, 6]. No simplifi- cation nor final...
-
Should Financial Institutions Engage in Interest Rate Swaps for Speculative Purposes?
-
How can poorly designed compensation schemes in financial services firms lead to conflicts of interest?
-
1-Stern observed all of the following results EXCEPT _______ in his experiment. A-one of the recombinant phenotypes was associated with an X chromosome of normal length B-the number of car, B+ male...
-
The greatest bond length is found in (a) O 2 ; (b) N 2 ; (c) Br 2 ; (d) BrCl.
-
When acrolein (propenal) reacts with hydrazine, the product is a dihydropyrazole: Suggest a mechanism that explains this reaction. H + H2N-NH2 Acrolein Hydrazine A dihydropyrazole
-
(a) Propose step-by-step mechanisms for both transformations of the Robinson annulations sequence just shown. (b) Would you expect 2-methylcyclohexane-1, 3-dione to be more or less acidic than...
-
Outline reasonable mechanisms that account for the products of the following Mannich reactions: (a) (b) (c) NMe2 Me2N NMe2 + 2 Me2NH CH3 CH
-
Multiplication of signed operands, which generate a double-length product in the 2's-complement 100 11 MULTIPLICAND (-13) 0 10 1 1 MULTIPLIER (+11) number system. The general strategy is the...
-
panel = Drawing Panel (160, 160) for i in range (0, 10): panel.draw rectangle (20, 20 + 100 10 * 1, 10 i, 10) Write variations of the above program that draw the figures at the lower right as output....
-
John estimated the following cash flows (in $) for a project: A B 1 Year Cash flow 2 0 -5,700 3 1 1,325 4 2 2,148 5 3 3,528 The required return for the project is 8%. What is the IRR for the project?

Study smarter with the SolutionInn App


