Calculate the potential energy of attraction between the Na + and Cl ? ions at the equilibrium
Question:
Calculate the potential energy of attraction between the Na+ and Cl? ions at the equilibrium separation r0 = 0.236 nm and compare this result with the dissociation energy given in Figure. What is the energy due to repulsion of the ions at the equilibrium separation?
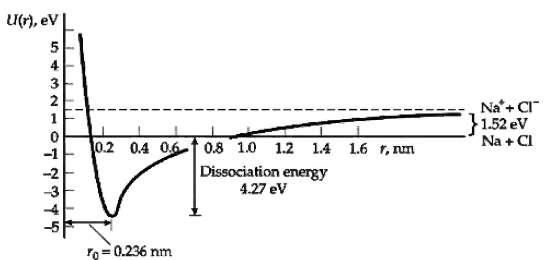
Transcribed Image Text:
U(r), ev 3 2 Na'+ CI" }1.52 ev Na + CI 0.2 0.4 06-f 0.8 1.0 1.2 14 1.6 1, nm Dissociation energy 4.27 ev = 0.236 nm
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 76% (17 reviews)
U e ke 2 r U e 1440236 eV 610 eV Dissociation ene...View the full answer

Answered By

Branice Buyengo Ajevi
I have been teaching for the last 5 years which has strengthened my interaction with students of different level.
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Fundamentals of Ethics for Scientists and Engineers
ISBN: 978-0195134889
1st Edition
Authors: Edmund G. Seebauer, Robert L. Barry
Question Posted:
Students also viewed these Modern Physics questions
-
FIGURE Q25.2 shows the potential energy of a proton (q = +e) and a lead nucleus (q = +82e). The horizontal scale is in units of femtometers, where 1 fm = 10 -15 m. a. A proton is fired toward a lead...
-
The potential energy of a diatomic molecule (a two-atom system like H2 or O) is given by where r is the separation of the two atoms of the molecule and A and B are positive constants. This potential...
-
The potential energy of a particle constrained to move along the x -axis is shown in the graph. At x = 0, the particle is moving in the + x -direction with a kinetic energy of 400 J. Can this...
-
1.1. Define a want and a need, and give an example of each. Then, explain how the relationship between wants and needs affects the way people spend money. (4 points) 2.. Identify and describe an...
-
Fabio Company uses a voucher system. Record the following transactions in the voucher register: 201X June 8 Purchased office equipment on account from Tiani Corporation, $1,200; voucher no. 300 was...
-
What are the major differences between a compilation engagement and a preparation engagement?
-
FHA Loan Company has 10,000 shares of \($3.50,\) no-par preferred stock and 50,000 shares of no-par common stock outstanding. FHA declared and paid the following dividends during a three-year period:...
-
Intuit Inc. develops and sells software products for the personal finance market, including popular titles such as Quicken and TurboTax. Classify each of the following costs and expenses for this...
-
The Joan Company uses the process cost system and average cost method. The following production data are for the month of July, 20--. Production Costs Work in Process beginning of the month:...
-
Marc Smith, vice president for operations of HHN, Inc., a manufacturer of cabinets for telephone switches, is constrained from meeting the 5-year forecast by limited capacity at the existing three...
-
Indicate the mean value of r for two vibration levels in the potential-energy curve for a diatomic molecule and show that because of the asymmetry in the curve, rav increases with increasing...
-
The equilibrium separation of the K + and F ions in KF is about 0.217 nm. (a) Calculate the potential energy of attraction of the ions, assuming them to be point charges at this separation. (b) The...
-
The Minneapolis-based medical instruments firm is considering starting an assembly operation in Bratislava (Slovakia) and is hesitating between establishing it as a branch or a subsidiary. The...
-
Which of the following best describes the level of measurement of the data listed in Exercise 1: nominal, ordinal, interval, ratio? Exercise 1 Literary Digest magazine conducted a poll for the 1936...
-
The numbers of the current players for the Chicago Bulls basketball team are 1, 2, 3, 5, 6, 9, 11, 13, 16, 20, 22, 26, 32, and 40. Does it make sense to calculate the average (mean) of these numbers?
-
The listed pulse rates were obtained for the Third National Health and Nutrition Examination Survey conducted by the U.S. Department of Health and Human Services, National Center for Health...
-
Table 2-2 on page 45 is a frequency distribution summarizing the IQ scores of a group of children with low lead exposure. Is it possible to identify the original list of the 78 individual IQ scores...
-
Survey Identify the type of sampling (random, systematic, convenience, stratified, cluster) used when a sample of the 1500 survey responses is obtained as described. Then determine whether the...
-
How do the cash management practices of large and small businesses differ?
-
Write each fraction as a percent. 7 50
-
_______ contraction drives blood through the systemic and pulmonary circuits; outside the heart, blood pressure is highest in the _______. a. Atrial; ventricles b. Atrial; atria c. Ventricular;...
-
In attempting to discern distant details, people will sometimes squint. Why does this help?
-
Is the image formed on the retina of the human eye upright or inverted? Discuss the implications of this for our perception of objects.
-
The human eye is much like a camera- yet, when a camera shutter is left open and the camera is moved, the image will be blurred; but when you move your head with your eyes open, you still see...
-
The normal freezing point of ethanol (CH3CH2OH) is -117.30 C and its Kfp value is 1.99 C/m. A nonvolatile, nonelectrolyte that dissolves in ethanol is testosterone . How many grams of testosterone,...
-
Farmhill Ltd . had 1 , 5 6 5 , 3 0 0 common shares outstanding on 1 January 2 0 X 6 , the beginning of its 2 0 X 6 fiscal year. During the year, on 1 May, the company issued 5 0 9 , 0 0 0 preferred...
-
Be sure to answer all parts. Balance the following equations and write the corresponding ionic and net ionic equations (if appropriate). Include phase abbreviations: Part 1 out of 2 (a) Provide the...

Study smarter with the SolutionInn App


