Calculate the total charge due to the charge distributions labeled A, B, C infigure. x=-2 P.=I mC/m
Question:
Calculate the total charge due to the charge distributions labeled A, B, C infigure.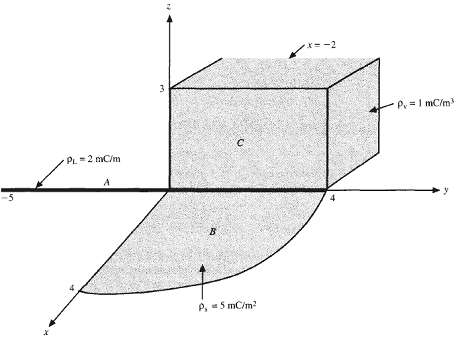
Transcribed Image Text:
x=-2 P.=I mC/m PL 2 mC/m -5 P. 5 mC/m?
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (8 reviews)
Q fpdl p dl 5p 10 mC S ...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Electrodynamics questions
-
Due to the charge distribution labeled B in figure,(a) Find E at point (0, 0, 3) if ?S = 5 mC/m2.(b) Find E at point (0, 0, 3) if ?S = 5 sin ? mC/m2. x=-2 P.=I mC/m PL 2 mC/m -5 P. 5 mC/m?
-
A total charge of 7.50 106 C is distributed on two different small metal spheres. When the spheres are 6.00 cm apart, they each feel a repulsive force of 20.0 N. How much charge is on each sphere?
-
A point charge Q = +4.60 C is held fixed at the origin. A second point charge q = + 1.20 C with mass of 2.80 X 10-4 kg is placed on the x-rods, 0.250 m from the origin. (a) What is the electric...
-
Write out the equation for the following chemical reaction and balance it. Solid potassium chlorate, KCIO3, decomposes to form solid potassium chloride and diatomic oxygen gas. What is the...
-
Why might Fed policymakers, in turn, experience difficulties determining which of the public's inflation expectations are the best signals of inflationary pressures in the economy?
-
An Online Food Ordering System allows the user of a web browser to order food for home delivery. To place an order, a shopper searches to find items to purchase, adds items one at a time to a...
-
The widely used engines for earth-moving machines are : (a) petrol engines (b) steam engines (c) diesel engines (d) gasoline engines
-
If a manufacturer sells its laundry detergent to a wholesaler for $2.50, at what price will the wholesaler sell it to a retailer if the wholesaler wants a 15 percent margin based on the selling...
-
Marvin Services Corporation had the following accounts and balances: Notes Payable ? Equipment $11,000 Accounts receivable $3,000 Land $31,000 Buildings $50,000 Total stockholders' equity $80,000...
-
1. Using the spreadsheet model from Case 2.1 as a starting point, use Solver to find the optimal set of projects to approve. The solution should maximize the total NPV from the approved projects, and...
-
Determine the total charge (a) On line 0 < x < 5 m if pz = 12x mC/m (b) On the cylinder p = 3,0 < z < < 4 m if es = pz2 nC/m? (c) Within the sphere r = 4 m if p, %3D C/m r sin 0 10
-
Find E at (5, 0, 0) due to charge distribution labeled A infigure. x=-2 P.=I mC/m PL 2 mC/m -5 P. 5 mC/m?
-
What strategic issues have the potential to create environmental uncertainty in the following four industries: (a) Automobile; (b) Social media; (c) Newspaper; (d) Medical services?
-
Its a sad yet heartwarming story. In 1998, Stephen Heywood, a strong 28-year-old carpenter who was building his dream house in California, got the worst possible news. He was diagnosed with...
-
In early 2008, Jason Jacobs was actively looking for a business idea to pursue. He knew he wanted to start his own businesshe just didnt know what that business would be. Anticipating the need for...
-
A friend of yours just bought a Samsung smartphone equipped with the Android operating system. While showing it to you, he said, You think entrepreneurial firms are so smart, look at what Samsung has...
-
Jacob Lacy is an undergraduate student at a Big 12 school. He has an idea to start an Internet-based firm that will help high school students prepare for college entrance exams. Jacob just talked to...
-
You just made a trip home and are visiting with your dad. He is 59 years old and has spent the past 12 years working in various management positions for Target. Prior to that, he served 20 years in...
-
As in Problem 9, consider a planar system of three identical masses and three identical springs. At equilibrium each mass is at the apex of an equilateral triangle. A spring links each pair of...
-
Marc Company assembles products from a group of interconnecting parts. The company produces some of the parts and buys some from outside vendors. The vendor for Part X has just increased its price by...
-
According to the experimental results in Eq. 7.33, is the addition of bromine to cis-2-butene a syn- or an anti-addition? H3C-CH=CH-CH3 cis trans Br CHCl2 Br Br 1 T H3C-CH-CH-CH3 racemate meso (7.33)
-
Explain the reasons for the projected increase in the U.S. budget deficit in coming years.
-
Why is the current path of fiscal policy not sustainable?
-
List the options for making fiscal policy sustainable.
-
Consider the seventh excited level of the hydrogen atom. A)What is the energy of this level? B)What is the largest magnitude of the orbital angular momentum? C)What is the largest angle between the...
-
1. How many degrees Celsius does the temperature decrease when there is a 45.8F decrease in temperature? 2. For each 1.00F change in temperature what is the corresponding change in degrees Celsius?...
-
A proton (mass of 1.67 x 10-27 kg) is moving at 1.25 x 106 m/s directly toward a stationary helium nucleus (mass 6.64 x 10-27 kg). After a head-on elastic collision, what is the velocity of the...

Study smarter with the SolutionInn App


