Carboxylic acids (RCO 2 H) react with alcohols (R?OH) in the presence of an acid catalyst. The
Question:
Carboxylic acids (RCO2H) react with alcohols (R?OH) in the presence of an acid catalyst. The reaction product of propanoic acid with methanol has the following spectroscopic properties. Propose a structure.
MS: M+ = 88
IR: 1735 cm?1
1H NMR: 1.11 ? (3 H, triplet, J = 7 Hz); 2.32 ? (2 H, quartet, J = 7 Hz); 3.65 ? (3 H, singlet)?
13C NMR: 9.3, 27.6, 51.4, 174.6 ?
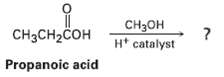
Transcribed Image Text:
CHзон H* catalyst CH3CH2COH Propanoic acid
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 81% (11 reviews)
The product M 88 has the formula C4H...View the full answer

Answered By

Aysha Ali
my name is ayesha ali. i have done my matriculation in science topics with a+ . then i got admission in the field of computer science and technology in punjab college, lahore. i have passed my final examination of college with a+ also. after that, i got admission in the biggest university of pakistan which is university of the punjab. i am studying business and information technology in my university. i always stand first in my class. i am very brilliant client. my experts always appreciate my work. my projects are very popular in my university because i always complete my work with extreme devotion. i have a great knowledge about all major science topics. science topics always remain my favorite topics. i am also a home expert. i teach many clients at my home ranging from pre-school level to university level. my clients always show excellent result. i am expert in writing essays, reports, speeches, researches and all type of projects. i also have a vast knowledge about business, marketing, cost accounting and finance. i am also expert in making presentations on powerpoint and microsoft word. if you need any sort of help in any topic, please dont hesitate to consult with me. i will provide you the best work at a very reasonable price. i am quality oriented and i have 5 year experience in the following field.
matriculation in science topics; inter in computer science; bachelors in business and information technology
_embed src=http://www.clocklink.com/clocks/0018-orange.swf?timezone=usa_albany& width=200 height=200 wmode=transparent type=application/x-shockwave-flash_
4.40+
11+ Reviews
14+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
A carbohydrate (S) decomposes in the presence of an enzyme (E) The Michaelis-Menten kinetic parameters were found to be as follows M 200 mol m 3 100 mol m 3 min a Calculate the change of substrate...
-
In the presence of an acid catalyst, acetaldehyde forms a trimer known as paraldehyde. Because it induces sleep when it is administered to animals in large doses, paraldehyde is used as a sedative or...
-
On heating 1,2,4-butanetriol in the presence of an acid catalyst, a cyclic ether of molecular formula C4H8O2 was obtained in 81-88% yield. Suggest a reasonable structure for this product.
-
Adel runs a construction equipment rental company, renting out loaders, generators, lifts and similar equipment on short term rentals. In the past, Adel has purchased the equipment he then rents out,...
-
A firm produces two products, X and Y. The production technology displays the following costs, where C (i, j) represents the cost of producing i units of X and j units of Y: C (0, 50) = 100 C (5, 0)...
-
A balanced delta-connected load is supplied by a 60-Hz three-phase source with a line voltage of 240 V. Each load phase draws 6 kW at a lagging power factor of 0.8. Find: (a) The load impedance per...
-
Consider the tPA process reactor (cultivation) section in Figure 2.14 (a): (a) For the third cultivator, change the reaction time to three days. To obtain a cell concentration of \(3 \times 10^{6}...
-
Calculate the cash dividends required to be paid for each of the following preferred stock issues: Required: a. The semiannual dividend on 6% cumulative preferred, $50 par value, 30,000 shares...
-
Assuming a 12% annual interest rate, determine the present value of a five-period annual annuity of $6.200 under each of the following situations: Note: Use tables, Excel, or a financial calculator....
-
In about 300 words, explain how incubators and accelerators work. In your answer, compare these two approaches and provide at least one example of each.
-
A 13C NMR spectrum of commercially available 2, 4-pentanediol, shows five peaks at 23.3, 23.9, 46.5, 64.8, and 68.1 ?. Explain. CHCH-CHCH3 2,4-Pentanediol
-
Nitrites (RC ? N) react with Grignard reagents (R'MgBr). The reaction product from 2-methyipropanenitrile with methyl magnesium bromide has the following spectroscopic properties. Propose a...
-
How can a market researcher make valuable use of social media content?
-
You are Assistant Manager of Royal Bank of Canada. Your bank recently changed policy in regard to dressing in the bank. With effect from April 1, 2021 only formal clothing is allowed from Monday to...
-
SYDNEY Account Help board ProctorU Help centre SONY is considering to set up a plant in the city of Shenchen to produce its Smart LED TV in China You are given the following facts about the...
-
Akiak Nori was raised in Arviat, Nunavut, and then attended a career school in Thunder Bay, Ontario, majoring in electronic technology. Approaching graduation, he sorted through dozens of job offers...
-
1) Microsoft Outlook 2) Microsoft Teams 3) Whatsapp/Phone massager 4) Google Gmail 5) Google Meet 6) Google Chat and many more with above mentioned, how employees use digital communication to...
-
This table "Transaction Entry" keeps each transaction made by each sales representatives. The SalesPrice and UnitCost are both for per unit of goods. Use the table "UnitCost" to Complete the Column...
-
a. Express the signal in terms of unit-step functions. b. Find the Laplace transform of the expression in (a) by using the shift on t -axis. \(g(t)= 0 if t <0 1-t if 0 1
-
By referring to Figure 13.18, determine the mass of each of the following salts required to form a saturated solution in 250 g of water at 30 oC: (a) KClO3, (b) Pb(NO3)2, (c) Ce2(SO4)3.
-
For each compound, would you expect greater solubility in water or in hexane? Indicate the kinds of intermolecular forces that occur between the solute and the solvent in which the molecule is most...
-
Using the information in Figure 16-2, calculate the values of Ho for the following reactions: In Figure 16.2 (a) (b) (c) (-359 predicted) (-240 predicted) resonance enerty encrgy energy 240 kJ/mol232...
-
Borazole, B3N3H6 is an unusually stable cyclic compound. Propose a structure for borazole, and explain why it is aromatic.
-
(a) Draw all the Kekulé structures of anthracene and phenanthrene. (b) Propose mechanisms for the two additions shown. (c) Most of the additions of bromine to double bonds gave entirely anti...
-
This project requires an investment in fixed assets, at time 0, of 44,000,000. Assets will be depreciated straight line in 5 years. This project has a 4 year life. The revenues for each of the 4...
-
Lorenzo Company applies overhead to jobs on the basis of direct materials cost. At year - end, the Work in Process Inventory account shows the following. Work in Process Inventory Date Explanation...
-
T-bill with a $10,000 par has 174 days until maturity. The T-bill has a bank discount bid quote of 2.208% and ask quote of 2.198%. What is the return on the investment in this T-bill if the trader...

Study smarter with the SolutionInn App


