Question: Nitrites (RC ? N) react with Grignard reagents (R'MgBr). The reaction product from 2-methyipropanenitrile with methyl magnesium bromide has the following spectroscopic properties. Propose a
Nitrites (RC ? N) react with Grignard reagents (R'MgBr). The reaction product from 2-methyipropanenitrile with methyl magnesium bromide has the following spectroscopic properties. Propose a structure.
MS: M+ = 86
IR: 1715 cm?1?
1H NMR: 1.05 ? (6 H, doublet, J = 7 Hz); 2.12 ? (3 H, singlet); 2.67 ? (1 H, septet, J= 7Hz)
13C NMR: 18.2, 27.2, 41.6, 211.2 ?
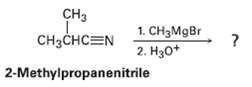
CH3 CHCEN 1. CH3MGB 2. * 2-Methylpropanenitrile
Step by Step Solution
3.37 Rating (166 Votes )
There are 3 Steps involved in it
The product is ... View full answer

Get step-by-step solutions from verified subject matter experts
Document Format (1 attachment)
22-C-O-S (240).docx
120 KBs Word File


