Complete the reactions given in Fig. P26.61, assuming the amino acid residue is part of a peptide
Question:
Complete the reactions given in Fig. P26.61, assuming the amino acid residue is part of a peptide in aqueous solution and is at neither the amino nor the carboxy terminus.
(a)
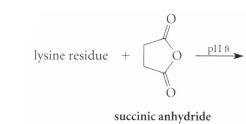
(b)
Transcribed Image Text:
plI 8 lysine residue + succinic anhydride
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (16 reviews)
a The sidechain amino group of lysine serves as a nucleophile to ...View the full answer

Answered By

Shadrack Mulunga
I am a Biochemistry by profession. However, I have explored different fields of study. My quest to explore new fields has helped me gain new knowledge and skills in Business, clinical psychology, sociology, organizational behavior and general management, and Project Management. I count my expertise in Project management, in particular, creation of Work Break Down Structure (WBS) and use of Microsoft Project software as one of my greatest achievement in Freelancing industry. I have helped thousands of BSC and MSC students to complete their projects on time and cost-effectively using the MS Project tool. Generally, I find happiness in translating my knowledge and expertise to success of my clients. So far, i have helped thousands of students to not only complete their projects in time but also receive high grades in their respective courses. Quality and timely delivery are the two key aspects that define my work. All those who hired my services always come back for my service. If you hire my services today, you will surely return for more. Try me today!
5.00+
154+ Reviews
289+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Using known reactions and mechanisms discussed in the text, complete the reactions given in Fig. P19.46 on p. 942. Fig. P19.46 NaBH4 CH OH
-
Complete the reactions given in Fig. P21.52 by giving the principal organic products. Explain how you arrived at your answers. NaOH CH O (trace) H,C CCHO CH +CH OH (solvent) Ph NH2 1 (CgH,NO3)...
-
Complete the reactions given in Fig. P22.81 by giving the major organic products. Explain your reasoning. NaOEt excess) EtOH H,o heat CHsI (c) CI NO, H ot, heat C CH LiAIH 2) HaO (e) CH Et CHCHCOEOH...
-
As programs become more complex, it becomes increasingly important to plan or "design" your code before writing it. Designing your code will help you organize its logic. It will also help you keep...
-
Predator Pucks, Inc. has current assets of $8,000, net fixed assets of $45,000, current liabilities of $6,800, and long-term debt of $13,800. What is the value of the shareholders' equity account for...
-
What would be the impact upon the consolidated financial statements if the dividend had been paid before the end of the subsidiarys accounting period?
-
Why should we think in terms of "failing to reject" the null rather than just accepting it?
-
The following transactions of Concerto Music Company occurred during 2014 and 2015: 2014 Mar 3 Purchased a piano (inventory) for $72,000, signing a six-month, 4% note payable. May 31 Borrowed $85,000...
-
A long straight current-carrying wire runs from north to south. A compass needle is placed above the wire points with its N-pole toward the east. In what direction is the current flowing? If a...
-
Hotel Baroneii International is an international hospitality group that is present in more than 100 countries with 12 brands and 5000 plus properties. The group also has a strong loyalty membership...
-
Draw the structure of the major neutral form of each of the following peptides, G-D-G-L-F
-
Show how the acetamidomalonate method can be used to prepare the following unusual amino acids from the indicated starting material and any other reagents. (a) (b) (c) (CH) CDCH CH CO from...
-
Find the mean and standard deviation of in Exercise 4.17. x p(x) 2 .. 1/36 3 .. 2/36 4 .. 3/36 5 .. 4/36 6 .. 5/36 7 .. 6/36 8 .. 5/36 9 .. 4/36 10 . 3/36 11 . 2/36 12 . 1/36
-
Suppose you have a circuit in which you have connected 2 batteries, each of which that has voltage of 2.8V, in series with each other such that the positive terminal of one battery is connected to...
-
Consider the following equivalent expressions: 3 133 and and 3/13 20 Enter the value of a followed by the value of b, separated by a comma. What are the values of a and b?
-
Simplify /45xy to the form a . V
-
concider a lithium - ion battery ( battery A ) that deliver's an energy of ( 5 0 wh ) . What is the Voltage al which the battery shoull be maintained duing the difctonge if it has a capacity of 2 5...
-
Factor by grouping. 5v +7v - 10v-14 6
-
Refer to the gravity equation. a. Why is trade greater between two large trading partners? b. How does distance between trading partners influence the amount of trade?
-
5. How much would you need to deposit in an account now in order to have $5,000 in the account in 5 years? Assume the account earns 2% interest compounded monthly. 10. You deposit $300 each month...
-
Propose structures for alcohols that have the following 1H NMR spectra: (a) C9H12O (b)C8H10O2 Part (a) TMS 10 O ppm Chemical shift (8) Part (b) TMS O ppm 10 8. Chemical shift (8) Intensity Intensity...
-
Compound A, C8H10O, has the IR and 1H NMR spectra shown. Propose a structure consistent with the observed spectra, and assign each peak in the NMR spectrum. Note that the absorption at 5.5 ?...
-
Propose a structure for a compound C15H24O that has the following 1H NMR spectrum. The peak marked by an asterisk disappears when D2O is added to thesample. TMS 10 8. 0 ppm Chemical shift (8)...
-
Townsend Produce agrees to sell 100 crates of lettuce to Metro Grocers under a shipment contract. Townsend arranges for Excel Transport to pick up and deliver the lettuce, which Excel Transport does....
-
Assume an organic compound has a partition coefficient between water and ethyl acetate equal to 8.12. If there are initially 7.10 grams of the compound dissolved in 75.0 mL of water, how many grams...
-
NO. DATE Use the table below Write a Java program that will display the output below. code may vary... For your reference QUANTITY ITEM PRICE 1 Bond Paper (rim) 230 1 Notebook 30 1 1 Printer Ink 5500...

Study smarter with the SolutionInn App


