Question: Show how the acetamidomalonate method can be used to prepare the following unusual amino acids from the indicated starting material and any other reagents. (a)
Show how the acetamidomalonate method can be used to prepare the following unusual amino acids from the indicated starting material and any other reagents.
(a)
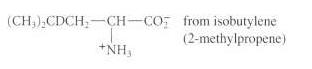
(b)
(c)
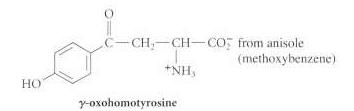
(CH) CDCH CH CO from isobutylene (2-methylpropene) +NHs fromn benzaldehyde Ph-CHD-CH-CO; NH C-CH.-CH-CO2 from anisole (methoxybenzene) NH HO -oxohomotyrosine
Step by Step Solution
3.47 Rating (173 Votes )
There are 3 Steps involved in it
Every synthesis has the same final steps The challenge in each case ... View full answer

Get step-by-step solutions from verified subject matter experts
Document Format (1 attachment)
902-O-EE (416).docx
120 KBs Word File


