Compute the atomic packing factor for cesium chloride using the ionic radii in Table 12.3 and assuming
Question:
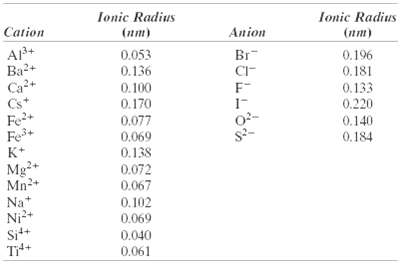
Compute the atomic packing factor for cesium chloride using the ionic radii in Table 12.3 and assuming that the ions touch along the cubediagonals.
Transcribed Image Text:
Ionic Radius Ionic Radius Cation (nm) Anion (nm) A3+ Ba2+ Ca2+ Cs* 0.053 Br 0.196 0.136 CI- 0.181 0.133 0.220 0. 100 0.170 Fe+ Fe+ K+ 0.077 0.069 0.138 0.140 0.184 Mg Mn2+ Na 0.072 0.067 * Nj2+ Si++ Tit+ 0.102 0.069 0.040 0.061
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (14 reviews)
We are asked in this problem to compute the atomic packing ...View the full answer

Answered By

Shaira grace
I have experience of more than ten years in handing academic tasks and assisting students to handle academic challenges. My level of education and expertise allows me communicate eloquently with clients and therefore understanding their nature and solving it successfully.
5.00+
4+ Reviews
10+ Question Solved
Related Book For 

Fundamentals of Materials Science and Engineering An Integrated Approach
ISBN: 978-1118061602
4th Edition
Authors: David G. Rethwisch
Question Posted:
Students also viewed these Materials Science Engineering questions
-
Compute the atomic packing factor for the diamond cubic crystal structure (Figure 12.15). Assume that bonding atoms touch one another, that the angle between adjacent bonds is 109.5, and that each...
-
Compute the atomic packing factor for the rock salt crystal structure in which rC/rA = 0.414.
-
Compute the atomic packing factor for the diamond cubic crystal structure (Figure 12.15). Assume that bonding atoms touch one another, that the angle between adjacent bonds is 109.5, and that each...
-
As an HR director for IMF Plastics Manufacturing, you receive a phone call about communicating changes made by the company to provide additional shifts.As soon as the call ends, you begin the process...
-
Briefly discuss about Employee Behavior and Organizational Design in the light of the following scenario. None of the factors that influence employee behavior exist in a vacuum. Spector (2013) says...
-
William Giese operates the Giese Broadcast Company. His postclosing trial balance on December 31, 2016, is as follows: Giese agrees to enter into a partnership with Hazel Borris, effective January 1,...
-
Within the revenue /receipt cycle, several paper and computer image documents are typically used to create an accounting system and, correspondingly, an audit trail for credit sales and cash...
-
Archer Construction Company began work on a $420,000 construction contract in 2010. During 2010, Archer incurred costs of $278,000, billed its customer for $215,000, and collected $175,000. At...
-
Explain how social media and related technology can be used to gather data and engage customers in the hotel and hospitality arena . Identify risks and gains. To complete your discussion, define what...
-
Determine the HC and CN bond lengths in HCN from the rotational constants B( 1 H 12 C 14 N) = 44.316 GHz and B( 2 H 12 C 14 N) = 36.208 GHz.
-
The unit cell for Cr2O3 has hexagonal symmetry with lattice parameters a = 0.4961 nm and c = 1.360 nm. If the density of this material is 5.22 g/cm3, calculate its atomic packing factor. For this...
-
For each of the following crystal structures, represent the indicated plane in the manner of Figures 3.11 and 3.12, showing both anions and cations: (100) plane for the rock salt crystal structure,...
-
If cable CB is subjected to a tension that is twice that of cable CA, determine the angle for equilibrium of the 10-kg cylinder. Also, what are the tensions in wires andCB? 30
-
a. What are the three primary methods of cost allocation? b. What are the differences among them?
-
Describe the tactics that a salesperson may use to deal with price concerns of a prospect.
-
List all the possible means of communication that might replace travel and person-toperson sales calls.
-
Explain the concept of direct denial. How should a salesperson deal with this type of buyer concern and resistance?
-
What are the three major sections of the balance sheet?
-
Problem The Medco pharmaceutical company has just developed a new antibiotic. Two percent of children taking competing antibiotics experience headaches as a side effect. A researcher for the Food and...
-
A bar of a steel alloy that exhibits the stress-strain behavior shown in Figure 6.22 is subjected to a tensile load; the specimen is 375 mm (14.8 in.) long and has a square cross section 5.5 mm (0.22...
-
Nitrogen is the primary component of our atmosphere. It is also used as an inert reagent to fill containers of chemicals that might react with the oxygen in air. Draw a Lewis structure of nitrogen...
-
Describe an engineering application where the density of the selected material would be an important material consideration.
-
What are the standard units used to report stress and strain in the English system? In the metric or SI system?
-
Why might Young's modulus or stiffness be an important material property?
-
4.5. Multiplication in GF(24): Compute A(x)-B(x) mod P(x) in GF(24) using the irreducible polynomial P(x) = x++x+1. What is the influence of the choice of the reduction polynomial on the computation?...
-
Write a C++ program that calculates the volume of a cylinder. Define appropriate variables for the diameter and the height of the cylinder. Assuming the units of these variables are in inches,...
-
a. Show the d and values that result from running breadth-first search on the directed graph of Fig 1, using vertex A as the source. (Ascending Order) b. Show the d, f and values that result from...

Study smarter with the SolutionInn App


