Question: Explain why neopentane shows no molecular ion is its mass spectrum, Predict the structure and m/z for these base ion in its massspectrum. CH3 CH
Explain why neopentane shows no molecular ion is its mass spectrum, Predict the structure and m/z for these base ion in its massspectrum.
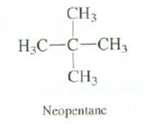
CH3 CH .-, -C- CH3 Neopentane
Step by Step Solution
★★★★★
3.45 Rating (165 Votes )
There are 3 Steps involved in it
1 Expert Approved Answer
Step: 1 Unlock

Neopentane does not show a molecular ion pea... View full answer

Question Has Been Solved by an Expert!
Get step-by-step solutions from verified subject matter experts
Step: 2 Unlock
Step: 3 Unlock
Document Format (1 attachment)
15-C-O-S (114).docx
120 KBs Word File


