Question: Determine values for the constants n and k (Equation 10.17) for the recrystallization of copper (Figure 10.11) at 102?C. y = 1 - exp(-kt) (10.17)
Determine values for the constants n and k (Equation 10.17) for the recrystallization of copper (Figure 10.11) at 102?C.
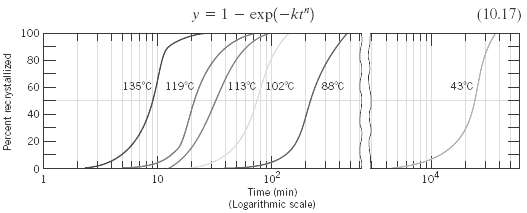
y = 1 - exp(-kt") (10.17) 100 80 113C 102 C 88C 135C/ 119C 43C 60 40 20 102 104 10 Time (min) (Logarithmic scale) Percent recrystallized
Step by Step Solution
3.47 Rating (173 Votes )
There are 3 Steps involved in it
In this problem we are asked to determine from Figure the values of the constants n and k Equation 1... View full answer

Get step-by-step solutions from verified subject matter experts
Document Format (1 attachment)
33-E-M-S-E-M-S (384).docx
120 KBs Word File


