Draw Fischer projections of the following molecules, placing the carbonyl group at the top in the usual
Question:
Draw Fischer projections of the following molecules, placing the carbonyl group at the top in the usual way. Identify each as a D or Lsugar.
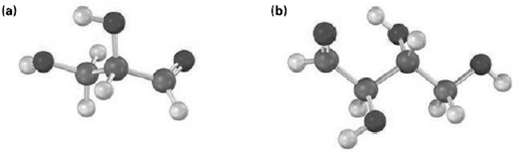
Transcribed Image Text:
(b) la)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 64% (14 reviews)
The hints in the previous proble...View the full answer

Answered By

Ali Khawaja
my expertise are as follows: financial accounting : - journal entries - financial statements including balance sheet, profit & loss account, cash flow statement & statement of changes in equity -consolidated statement of financial position. -ratio analysis -depreciation methods -accounting concepts -understanding and application of all international financial reporting standards (ifrs) -international accounting standards (ias) -etc business analysis : -business strategy -strategic choices -business processes -e-business -e-marketing -project management -finance -hrm financial management : -project appraisal -capital budgeting -net present value (npv) -internal rate of return (irr) -net present value(npv) -payback period -strategic position -strategic choices -information technology -project management -finance -human resource management auditing: -internal audit -external audit -substantive procedures -analytic procedures -designing and assessment of internal controls -developing the flow charts & data flow diagrams -audit reports -engagement letter -materiality economics: -micro -macro -game theory -econometric -mathematical application in economics -empirical macroeconomics -international trade -international political economy -monetary theory and policy -public economics ,business law, and all regarding commerce
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Draw Fischer projections of L-aspartate and L-cysteine and show why L-Asp is (S)-Asp and L-Cys is (R)-Cys.
-
Draw Fischer projections of L-glucose and L-fructose.
-
Draw Fischer projections of the following molecules: (a) The S enantiomer of 2-bromobutane (b) The R enantiomer of alanine, CH3CH (NH2) COOH (c) The R enantiomer of 2-hydroxypropanoic acid (d) The S...
-
The following data has been extracted from last quarter's budget of Elise Ltd, which manufactures and sells a single product. January February March Production units 6200 6200 5800 Sales units 5900...
-
Visit any restaurant, possibly your school cafeteria. The workers and fellow customers will assume that you are an ordinary customer, but you are really a spy for the owner. Your task After your...
-
Compute the density for nickel at 500C, given that its room-temperature density is 8.902 g/cm3. Assume that the volume coefficient of thermal expansion, v, is equal to 3l.
-
Refer to Example 11. Ignore the covariate original reflectivity. Perform an analysis of variance take \(\alpha=0.05\) Data From Example 11 EXAMPLE II Creating an analysis of covariance table with one...
-
Multiple Choice Questions The following questions concern internal controls in the inventory and warehousing cycle. Choose the best response. a. Which of the following controls will most likely...
-
Mexican Peso Finanicial Statements Assests: Non Curent Assets $ 35,000,000.00 Current Assests $ 5,000,000.00 Total Assests $ 40,000,000.00 Equity $ 25,000,000.00 Non Current Liabilities $...
-
On March 10, 2006, Nortel Networks Corp. announced that it would delay filing its 2005 financial reports with the SEC. The delay arose because Nortel and its auditors decided that certain revenue...
-
Identify the following aldoses, and tell whether each is a D or Lsugar: (a) (b)
-
The following structure is that of an L aldohexoses in its pyranose form. Identify it, and tell whether it is an ? a or ? anomer.
-
What distinguishes a current liability from a long-term liability? What distinguishes a contingent liability from an actual liability?
-
The Plaintiff Alisha is suing the Defendant Benadine in a civil action claiming damages in a court of competent jurisdiction in Pacifica arising out of a collision at an intersection on 6 th August...
-
When bart bought his car, it averaged 28 miles per gallon of gas. Now, the car's average miles per gallon has decreased by 14%. What is the cars average miles per gallon now?
-
Attorney Lillian Liberty wants to expand the firm's sports law practice, and is very pleased to have a new client, Dwayne Simpson, a waterpolo player. Liberty wants you to assist with Simpson's...
-
Using the articles provided in bold below what trends seem to emerge from the public health workforce literature over the last 10 years? What are the most critical issues facing the public health...
-
In 2006 Jack Kelly Partners LLC alleged that Zegelstein (defendants) advertised that the premises was suitable for commercial use. Plaintiff was not aware that the use as represented by defendants...
-
An investment has the following cash flow series where interest is 5 percent: a. Determine the present worth of the series. b. Determine the future worth of the series at the end of year 8. c. Find...
-
Reread the discussion leading to the result given in (7). Does the matrix sI - A always have an inverse? Discuss.
-
The initial concentration and K a of several weak acid (HA) solutions are listed. For which solution is the x is small approximation least likely to work in finding the pH? (a) Initial [HA] = 0.100...
-
Draw the structure of a. A primary alkyl chloride, C4H9Cl. b. A tertiary alkyl bromide, C5H11Br. c. A secondary alkyl iodide, C6H11I.
-
Using Table 6.1, write an equation for each of the following substitution reactions: a. p-methylbenzyl chloride + sodium acetylide b. n-propyl bromide + sodium cyanide c. 2-iodopropane + sodium...
-
Select an alkyl halide and a nucleophile that will give each of the following products: a. CH3OCH2CH2CH2CH3 b. HC¡CCH2CH2CH3 c. CH3CH2CH2NH2 d. CH3CH2CH2SCH2CH2CH3 e. f. CH2CN on OCH CH
-
MM Corp. has 50,000 shares outstanding with share price of $18. It has debt with market value of $300,000. The equity beta is 1.2 and debt beta is 0.1. The risk-free rate is 2% and the market risk...
-
The market is expected to return 15 percent next year and the risk-free rate is 7 percent. What is the expected rate of return on a stock with a beta of 1.3? The covariance of the market's returns...
-
A stock's current price is 145.05. A put option with an exercise price of 120 and maturity of 3 months is currently priced at $ 28.83. What is the option's time value?

Study smarter with the SolutionInn App


