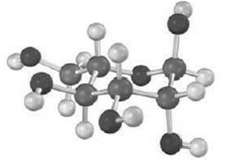
The following structure is that of an L aldohexoses in its pyranose form. Identify it, and tell
Question:
The following structure is that of an L aldohexoses in its pyranose form. Identify it, and tell whether it is an ? a or ? anomer.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (10 reviews)
The structure represents an o anomer because th...View the full answer

Answered By

Somshukla Chakraborty
I have a teaching experience of more than 4 years by now in diverse subjects like History,Geography,Political Science,Sociology,Business Enterprise,Economics,Environmental Management etc.I teach students from classes 9-12 and undergraduate students.I boards I handle are IB,IGCSE, state boards,ICSE, CBSE.I am passionate about teaching.Full satisfaction of the students is my main goal.
I have completed my graduation and master's in history from Jadavpur University Kolkata,India in 2012 and I have completed my B.Ed from the same University in 2013. I have taught in a reputed school of Kolkata (subjects-History,Geography,Civics,Political Science) from 2014-2016.I worked as a guest lecturer of history in a college of Kolkata for 2 years teaching students of 1st ,2nd and 3rd year. I taught Ancient and Modern Indian history there.I have taught in another school in Mohali,Punjab teaching students from classes 9-12.Presently I am working as an online tutor with concept tutors,Bangalore,India(Carve Niche Pvt.Ltd.) for the last 1year and also have been appointed as an online history tutor by Course Hero(California,U.S) and Vidyalai.com(Chennai,India).
4.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The following structure is drawn in an unconventional manner. (a) Label the N terminus and the C terminus. (b) Label the peptide bonds. (c) Identify and label each amino acid present. (d) Give the...
-
The following structure is an intermediate in a synthesis of prostaglandins F2a and E2 by E. J. Corey (Harvard University). A Horner-Wadsworth-Emmons reaction was used to form the (E)-alkene. Write...
-
The following 1H NMR spectrum is that of an alcohol, C8H10O. Propose a structure. TMS 6. Chemical shift (8) 3 O ppm 10 8. Intensity
-
Patients who undergo chronic hemodialysis often experience severe anxiety. Videotapes of progressive relaxation exercises were shown to one group of patients and neutral videotapes to another group....
-
The use of social networks by employees during work hours remains a controversial topic, with some companies encouraging networking, some at least allowing it, and others prohibiting it. Your task...
-
When a metal is heated its density decreases. There are two sources that give rise to this diminishment of : (1) the thermal expansion of the solid, and (2) the formation of vacancies (Section 4.2)....
-
To determine the effect of height on power generated in a hydroelectric power plant, the following observations were made:. Use the level of significance \(\alpha=0.05\) to test whether the height of...
-
A hotel housekeeper, Alison Harvey, was observed five time son each of four task elements, as shown in the following table. On the basis of these observations, find the standard time for the process....
-
The expected dividend per share on the equity share of Roadport Limited is $2.00. The dividend per share of Roadport Limited has grown over the past five years at the rate of 5% per year. This growth...
-
A dog training business began on December 1. The following transactions occurred during its first month. December 1 Receives $23,000 cash as an owner investment in exchange for common stock. December...
-
Draw Fischer projections of the following molecules, placing the carbonyl group at the top in the usual way. Identify each as a D or Lsugar. (b) la)
-
The following model is that of an aldohexose: (a) Draw Fischer projections of the sugar, its enantiomer, and a diastereomer. (b) Is this a D sugar or an L sugar? Explain. (c) Draw the ? anomer of the...
-
1. Was this a good use of the audit concept? 2. What was the major problem in Auditing a Troubled Project at Atlantic States Chemical Laboratories? 3. In spite of the recommendation, ASCL had already...
-
A sex offender is convicted and sentenced to a long jail term. Say 10 years. After serving their sentence rather than being released they are civilly committed and remain incarcerated at a mental...
-
Antonio believes he has been misled by his employer, MiraCosta College. He decides to sue. He knows you have recently taken BUS140 and asks what you think about going to court. Would you advise him...
-
Jamie Jones is a resident at Miller's Retirement Center. She has dementia. Some days she seems without ailment. Dr. Smith needs her permission for a minor surgery. Is there any way to get Ms. Jones'...
-
Harry lends his car to his friend Alice for the month of January while she is away on holiday overseas. a) Explain who the owner of the car is during this time. b) If Alice allows her younger sister...
-
Jeanne Dunne, a bank teller, provided a description of a bank robber "moments" following the robbery. The robber was identified as an African American with dark skin, a wide nose, and medium build....
-
The manager at a Sherwin-Williams store has decided to purchase a new \($30\),000 paint-mixing machine with hi-tech instrumentation for matching color and other components. The machine may be paid...
-
In the current year, the City of Omaha donates land worth $500,000 to Ace Corporation to induce it to locate in Omaha and create an estimated 2,000 jobs for its citizens. a. How much income, if any,...
-
Find the pH of a 0.350 M aqueous benzoic acid solution. For benzoic acid, K a = 6.5 * 10 -5 . a) 4.64 b) 4.19 c) 2.32 d) 11.68
-
Draw each of the following equations in a way that shows clearly the stereochemistry of the reactants and products. a. (R)-2-bromobutane sodium methoxide (in methanol)2-methoxybutane b....
-
The (+) enantiomer of the inhalation anesthetic desflurane (CF3CHFOCHF2) has the S configuration. Draw a three-dimensional representation of (S)-(+)-desflurane.
-
When treated with sodium iodide, a solution of (R)-2-iodooctane in acetone gradually loses all of its optical activity. Explain.
-
An investor put 60 percent of his money into a risky asset offering a 10 percent return with a standard deviation of return of 8 percent, and he put the balance of his risk-free asset offering 5...
-
There is a bonus with the following characteristics: nominal value $98.650.000 annual coupon rate 4.5% term 5 years, tir 6.5% A) Calculate the price of the bond assuming it is zero coupon B)...
-
Ida Company produces a handcrafted musical Instrument called a gamelan that Is similar to a xylophone. The gamelans are sold for $949. Selected data for the company's operations last year follow:...

Study smarter with the SolutionInn App


