Electrophilic addition of Br2 to isoprene (2-methyl-1, 3-hutadiene) yields the following product mixture: Of the 1, 2-addition
Question:
Electrophilic addition of Br2 to isoprene (2-methyl-1, 3-hutadiene) yields the following product mixture: Of the 1, 2-addition products, explain why 3, 4-dibromo-3-methyl-1-butene (21 %) predominates over 3, 4-dibromo-2-mcthyl-1-butene(3%).
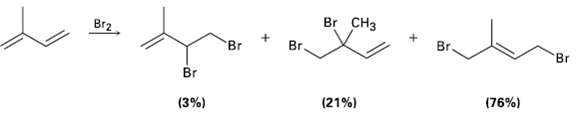
Transcribed Image Text:
Br CHз Br2 Br. Br Br 'Br Br (76%) (21%) (3%)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (9 reviews)
CH3 8 8 BrCH CCHCH A tertiaryprimary allylic carboca...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The rate law for addition of Br2 to an alkene is first order in Br2 and first order in the alkene. Does this fact prove that the mechanism of addition of Br2 to an alkene proceeds in the same manner...
-
Explain why methyl propyl ether forms both methyl iodide and propyl iodide when it is heated with excess HI.
-
Explain why the following reaction yields the Hofmann product exclusively (no Zaitsev product at all) even though the base is not sterically hindered: Br NaOEt ELOH
-
Recall the heat equation which we solved numerically T= = DTxx There we implemented an explicit numerical scheme (FTCS) which led to a conditionally stable solution - meaning that for certain time...
-
Would an employee who posts, "I hate my job!" be protected, according to the principles cited here? Why or why not?
-
Find Vx in the network shown in Fig. 13.78. 2 2 /4
-
Refer to Example 1 and the data on the resiliency modulus of recycled concrete. (a) Obtain a 95% confidence interval for the population mean resiliency modulus \(\mu\). (b) Is the population mean...
-
Mobility Partners makes wheelchairs and other assistive devices. For years it has made the rear wheel assembly for its wheelchairs. A local bicycle manufacturing firm, Trailblazers, Inc., offered to...
-
Assume the company's tax rate is 35 percent. Debt: 9,000 7 percent coupon bonds outstanding, $1,000 par value, 20 years to maturity, selling for 106 percent of par; the bonds make semiannual...
-
Lincoln Petroleum has the following data in connection with Lease A: REQUIRED: Lincoln Petroleum is sole owner of the working interest. If reserves are determined to be 11,000 barrels, is the well...
-
Treatment of 3, 4-dibromohexane with strong base leads to loss of 2 equivalents of HBr and formation of a product with formula C6H10. Three products are possible. Name each of the three, and tell how...
-
Propose a structure for a conjugated diene that gives the same product from both 1, 2- and 1, 4-addition of HBr.
-
Is the function even, odd, or neither? f(x) = x2 2x + 1
-
The telecommunication sector is made up of companies that make communication possible on a global scale, whether it is through the phone or the Internet, through airwaves or cables, through wires or...
-
Identify a real (you may visit and use one of the listed cases) or fictitious case with an underpinning problem and address the following below: https://www.pmi.org/business-solutions/case-studies...
-
Given the data below ,what should the total salesforce be for my brand in thailand ? Should i go for total 9 or 147 ? Is it wise to share salesforce total evenly to all channels? Brand Indie Drug...
-
Molander Corporation is a distributor of a sun umbrella used at resort hotels. Data concerning the next month s budget appear below: Selling price per unit $ 2 4 Variable expense per unit $ 1 5 Fixed...
-
Michael is a senior student on job market. He is interested in a social media management position at Facebook. So before the interview, he decides to manage his own profile on Facebook and on...
-
Evaluate \(x\left(0^{+} ight)\)by using the IVT. \(X(s)=\frac{s^{2}+1}{\left(\frac{1}{2} s+1 ight)\left(9 s^{2}+6 s+2 ight)}\)
-
Show that gj concave AHUCQ Abadie For nonnegative variables, we have the following corollary.
-
The enthalpy of solution for NaOH is -44.46 kJ/mol. What can you conclude about the relative magnitudes of the absolute values of Hsolute and Hhydration , where Hsolute is the heat associated with...
-
Predict the major products of the following reactions. (a) 2, 4-dinitrochlorobenzene + NaOCH3 (b) Phenol + tert-butyl chloride + AlCl3 (c) Nitrobenzene + fuming sulfuric acid (d) Nitrobenzene +...
-
Predict the major products of bromination of the following compounds, using Br2 and FeBr3 in the dark. (a) (b) (c) NO, OCH OCH3 OCH,
-
What products would you expect from the following coupling reactions? (a) (b) (c) (d) (e) Br 2 CuLi PdCl2 Pd catalyst, base OR OR Pd catalyst, base Pd OAc)2 PPh;
-
java code ReadFile Create-a linked list from an input file (input.txt) that contains an even number of first names. The number of items in the file is unknown. Split Create-a split function that...
-
Complete a method named findValue which accepts an IntArray and an Int value and returns whether the array contains the passed value. Your method should not modify the passed array. However, you...
-
Write program Sum.java that just prints the strings that it is given at the command line, one per line. If nothing is given at the command line, print "No arguments". Modify your program (Make a copy...

Study smarter with the SolutionInn App


