Explain why the presence of a triple bond is much easier to detect in the IR spectrum
Question:
Explain why the presence of a triple bond is much easier to detect in the IR spectrum of 1-hexyne than it is in the spectrum of3-hexyne.
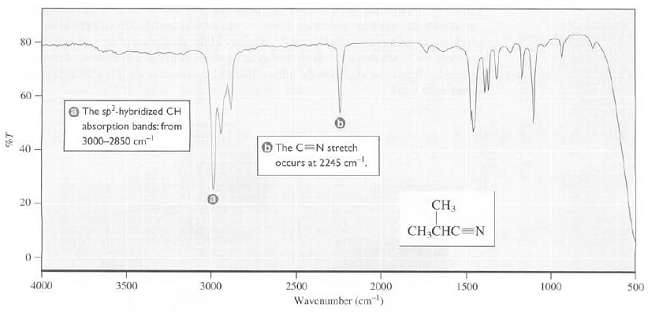
Transcribed Image Text:
80 60 O The sp-hybridized CH absorption bands: from 3000-2850 cm O The CN stretch occurs at 2245 cm. 40 20 CH CH,CHC=N 4000 3500 3000 2500 2000 1500 1000 500 Wavenumber (cm)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 61% (13 reviews)
The band in the triple bond region at 21502100 ...View the full answer

Answered By

Asim farooq
I have done MS finance and expertise in the field of Accounting, finance, cost accounting, security analysis and portfolio management and management, MS office is at my fingertips, I want my client to take advantage of my practical knowledge. I have been mentoring my client on a freelancer website from last two years, Currently I am working in Telecom company as a financial analyst and before that working as an accountant with Pepsi for one year. I also join a nonprofit organization as a finance assistant to my job duties are making payment to client after tax calculation, I have started my professional career from teaching I was teaching to a master's level student for two years in the evening.
My Expert Service
Financial accounting, Financial management, Cost accounting, Human resource management, Business communication and report writing. Financial accounting : • Journal entries • Financial statements including balance sheet, Profit & Loss account, Cash flow statement • Adjustment entries • Ratio analysis • Accounting concepts • Single entry accounting • Double entry accounting • Bills of exchange • Bank reconciliation statements Cost accounting : • Budgeting • Job order costing • Process costing • Cost of goods sold Financial management : • Capital budgeting • Net Present Value (NPV) • Internal Rate of Return (IRR) • Payback period • Discounted cash flows • Financial analysis • Capital assets pricing model • Simple interest, Compound interest & annuities
4.40+
65+ Reviews
86+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
A test for the presence of a certain disease has probability .20 of giving a false-positive reading (indicating that an individual has the disease when this is not the case) and probability .10 of...
-
Explain why the presence of a charismatic leader tends to enhance the job satisfaction of group members.
-
The shaft of a generator is much easier to turn when the generator is not connected to an outside circuit than when such a connection is made. Why?
-
Which power plant has high load factor?
-
The Stoney Company sells many products. Wolie is one of its popular items. Below is an analysis of the inventory purchases and sales of Wolie for the month of March. Stoney Company uses the periodic...
-
How would you assess the attractiveness of a new consumer product-market for Procter & Gamble? List the specific factors you would include as you build an index of market attractiveness for a Procter...
-
For a random sample of n = 25, find the probability of a sample mean being less than 100 or greater than 102 when = 100 and = 4.5. The population mean and standard deviation are given. Find the...
-
A company with no inventory buys the following three inventory items: On January 10, the company sells one item for $10. On January 15, the company sells a second item $10. The company uses a...
-
Gerald Sales has total owner's equity of $18,700. The firm has current liabilities of $2,500 and total assets of $31,800. What is the value of the long-term debt, if you also know that market value...
-
Meredith Ltd makes a special-purpose machine D4H used in the textile industry. Meredith has designed the D4H machine for 2018 to be distinct from its competitors. It has been generally regarded as a...
-
Indicate the positions of the absorption bands and any other noteworthy features in the hydrogen region of the IR spectra of thesecompounds: CH3 ) C,CH b) NH2 d) CH2=CHCH,CH,OH f) CH,CH,CH,CH e)...
-
The exhaust from a poorly maintained automobile may contain a wide variety of different hydrocarbon pollutants. Why is the 3000 to 2900 cm1 region a good place to monitor the amount of these...
-
The authors of the article Luck of the Draw: Creating Chinese Brand Names (J. of Advertising Res. 2008: 523530) counted the number of strokes in the characters for the names of 1202 Chinese brand...
-
In the calculation 4.56700 x 10-2 multiplied by 1.090 x 10-19, how many significant figures should you report in the answer? 04 01 02 3 05 Question 6 Which one of the following statements about...
-
Equivalent Taxable Yield rm = r(1-t) r = rm/(1-t) Example: Suppose your tax rate is 25% on a taxable corporate bond. A municipal bond is available paying 4% interest. What interest rate would you...
-
1. An amount of money, P, is invested in a simple savings account earning 4.25% annual interest. Assume interest is compounding continuously. a. How long will it take the amount of money to triple?...
-
A particular US-mined coal (typical gravimetric analysis: 80% C, 15% "ash", 3% S, 2% H) is burned completely with 120% theoretical air. Reactants are to be assumed to be at 25 C, 1 bar, and the...
-
Details of the capital structure of Webber Ltd. appear below: Bonds Number issued Coupon rate Interest payments Years to maturity 27,000 6% semi-annually 10 $90 Current price Preferred shares Number...
-
A dataset has 1000 records and 50 attributes with 5% of the values missing, spread randomly throughout the records and attributes. An analyst decides to remove records with missing values. About how...
-
A bar of length = 1 has one fixed and one free end and stiffness function c(x) = 1 - x. Find the displacement when subjected to a unit force. Pay careful attention to the boundary condition at the...
-
In 2008, the German-owned DHL Express decided that it could no longer compete with FedEx and UPS in the U.S. domestic package delivery market. The company had lost more than $1 billion a year in 2006...
-
Identify the monomer units from which each of the following polymers is made, and tell whether each is a chain-growth or a step-growthpolymer. (b) +CF2-CFCI (c) NHCH2CH2CH2C (a) +CH2-0+o (d) (e)
-
Draw a three-dimensional representation of segments of the following polymers: (a) Syndiotactic poly acrylonitrile (b) Atactic poly (methyl methacrylate) (c) Isotactic poly (vinyl chloride)
-
Draw the structure of Kodel, a polyester prepared by heating dimethyl 1, 4-henzcnedicarhoxylate with 1, 4-bis (hydroxymethyl)cyclohexane. H? -CH2 1,4-Bis(hydroxymethyl)cyclohexane
-
The partners mehak and simran share in the profits and losses equally and their capital accounts have credit balance s of 1 5 0 0 0 and 2 5 0 0 0 respectively. The account balances for Mehak and...
-
LNS Corporation reports book profit of $ 2 , 0 0 0 , 0 0 0 . The $ 2 , 0 0 0 , 0 0 0 included $ 1 5 , 0 0 0 of tax exempt interest income, a capital loss of $ 2 , 0 0 0 , depreciation expense of $ 6...
-
Maggie Vitteta, single, works 3 8 hours per week at $ 1 9 . 0 0 an hour. How much is taken out for federal income tax?

Study smarter with the SolutionInn App


