Identify the monomer units from which each of the following polymers is made, and tell whether each
Question:
Identify the monomer units from which each of the following polymers is made, and tell whether each is a chain-growth or a step-growthpolymer.
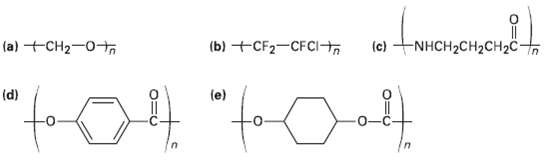
Transcribed Image Text:
(b) +CF2-CFCI (c) NHCH2CH2CH2C (a) +CH2-0+o (d) (e)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (12 reviews)
a n CO H c NH HNCHCHCHOH n d n HO e CH07 Chain...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Identify the statutory authority under which each of the following is exempt from Federal income tax. a. Kingsmill Country Club. b. Shady Lawn Cemetery. c. Peach Credit Union. d. Veterans of Foreign...
-
Identify the cycle to which each of the following general ledger accounts will ordinarily be assigned: sales, accounts payable, retained earnings, accounts receivable, inventory, and repairs and...
-
Identify the journal in which each of the following transactions is recorded. 1. Cash sales 2. Owner withdrawal of cash 3. Cash purchase of land 4. Credit sales 5. Purchase of merchandise on account...
-
M/s Active Builders Ltd. invested in the shares of another company (with an intention to hold the shares for short term period )on 31st October, 2016 at a cost of Rs.4,50,000. It also earlier...
-
What is the relationship between a firm's customers and its business-level strategy in terms of who, what, and how? Why is this relationship important?
-
Every other year, the National Institutes of Health publish the Report on Carcinogens, which lists products that cause cancer. Among those in the most recent report was formaldehyde, found in...
-
The table below shows the condition indices and variance decomposition proportions for the acetylene data using centered regressors. Use this information to diagnose multicollinearity in the data and...
-
Alpha Products, Inc., is having a problem trying to control inventory. There is insufi cient time to devote to all its items equally. Here is a sample of some items stocked, along with the annual...
-
Sweeten Company had no jobs in progress at the beginning of March and no beginning inventories. The company has two manufacturing departments-Molding and Fabrication. It started, completed, and sold...
-
1. What might Lewis have done to avoid the result in this case? Discuss. 2. Suppose that Lewis had already been reimbursed for the two vehicles that had been sold, and Whitney sought to obtain the...
-
Show the structures of the polymers that could be made from the following monomers (yellow-green =Cl): (b) (a)
-
Draw a three-dimensional representation of segments of the following polymers: (a) Syndiotactic poly acrylonitrile (b) Atactic poly (methyl methacrylate) (c) Isotactic poly (vinyl chloride)
-
In Exercises use a calculator to approximate the value. Round your answer to two decimal places. arcsec 1.269
-
If you find that you have income or deductions that you did not report on your tax return, you should:
-
U.S. GAAP includes these indicators as guidance for determining a subsidiary's functional currency, except for: Multiple Choice source of taxation. sources and uses of cash flows. sources of...
-
Assume that over the 30-years of retirement you are able to earn a weighted average of 7% compounded annually through investing in bonds, stock, and CD's. Using this rate, discount the total expenses...
-
Use information above to solve Survival Revenue, Average Selling price, and Number of units needed to sell
-
When a corporation sells its assets on the installment method prior to making a liquidation distribution of the installment notes, a shareholder receiving installment notes in a liquidation: Question...
-
Consider again the system of Fig. P4.4-5. Add a sampler for \(E(s)\) at the input. Given \[ G_{1}(s)=\frac{1}{s+10} \quad D(z)=\frac{z-0.5}{z-1} \quad G_{2}(s)=\frac{s}{s^{2}+9 s+23} \] find \(c(k...
-
Continuation of Exercise 4-83. (a) What is the probability that the first major crack occurs between 12 and 15 miles of the start of inspection? (b) What is the probability that there are no major...
-
Carbon disulfide is produced from the high-temperature reaction of carbon and sulfur: C(s) + S 2 (g) = CS 2 (g) This reaction is carried out in a retort at low pressure, and in the absence of oxygen...
-
Write a structural formula for each of the following compounds: (a) 6-Isopropyl-2,3-dimethylnonane (b) 4-tert-Butyl-3-methylheptane (c) 4-Isobutyl-1,1-dimethylcyclohexane (d) sec-Butylcycloheptane...
-
Give the IUPAC name for each of the following compounds: (a) CH3(CH2)25CH3 (b) (CH3)2CHCH2(CH2)14CH3 (c) (CH3CH2)3CCH(CH2CH3)2 (d) (e) (f) (g)
-
All the parts of this problem refer to the alkane having the carbon skeleton shown. (a) What is the molecular formula of this alkane? (b) What is its IUPAC name? (c) How many methyl groups are...
-
1. Electric potential is given by V=6x-8xy-8y+6yz - 4z Then magnitude of electric force acting on 2C point charge placed on origin will be :- (1) 2N (2) 6N (3) 8N (3) 8N (4) 20 N 2. Figure shows...
-
17. A concave mirror produces a real image 10mm tall, of an object 2.5mm tall placed at 5cm from the mirror. Calculate focal length of the mirror and the position of the image. 18. An object is...
-
23. A convex mirror used for rear view on an automobile has a radius of curvature of 3m. If a bus is located at 5m from this mirror, find the position, nature and magnification of the image. 24. An...

Study smarter with the SolutionInn App


