Indicate the positions of the absorption bands and any other noteworthy features in the hydrogen region of
Question:
Indicate the positions of the absorption bands and any other noteworthy features in the hydrogen region of the IR spectra of thesecompounds:
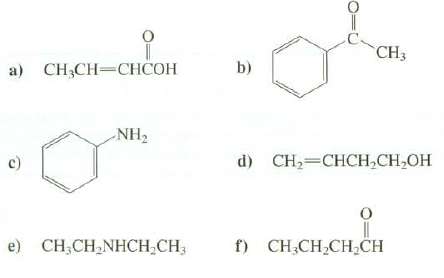
Transcribed Image Text:
CH3 а) CН,CH—СНСОН b) NH2 d) CH2=CHCH,CH,OH f) CH,CH,CH,CH e) CH;CH,NHCH;CH;
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 77% (9 reviews)
a OH 3000 cm 1 very broad CH 31003000 cm 1 CH 30002850 cm 1 ...View the full answer

Answered By

Ayush Jain
Subjects in which i am expert:
Computer Science :All subjects (Eg. Networking,Database ,Operating System,Information Security,)
Programming : C. C++, Python, Java, Machine Learning,Php
Android App Development, Xamarin, VS app development
Essay Writing
Research Paper
History, Management Subjects
Mathematics :Till Graduate Level
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Predict the positions of the absorption bands in the IR spectra for the carbonyl groups of thesecompounds. HO. b) CH,CH-CHCH (p
-
For the following reaction profiles, indicate The positions of reactants and products. The activation energy. ÎE for the reaction. The second reaction profile is representative of a reaction...
-
The IR and mass spectra for three different compounds are shown in Figures 13.45-13.47. Identify each compound. a. b. c. 25 26 2.7 28 19 2000 100 43 58 100 71 85 0 20 40 60 80 100 120 m/z 13 14 15 6...
-
Determine the number of valence electrons for each of thefollowing four elements. Part A Ga _______ Express your answer as an integer. Part B Pb ________ Express your answer as an integer. Part C Cl...
-
Chase Opportunity has selected you as the project manager to oversee their latest project - Chase Retail Complex. Chase's vision is to build four (4) 10,000 square foot buildings (see concept drawing...
-
Why would a business like Coca-Cola opt to use the GE/McKinsey portfolio model over other portfolio models?
-
Repeat Exercise 20 for samples of size 18 and 12. What happens to the mean and the standard deviation of the distribution of sample means as the sample size decreases? Data from Exercises 20 The...
-
A company that we call DC is a Fortune 100 diversified conglomerate with operations in many industries around the world. Top management focuses on the annual earnings in evaluating the performance of...
-
Pro, Inc. has just paid a dividend of $0.55 per share. The dividends are expected to grow at an annual rate of 5 percent indefinitely. What is the stock price today if the required return is 12.5...
-
The FASB ASC paragraph 810-10-45-16 states: "The non-controlling interest shall be reported in the consolidated statement of financial position within equity, separately from the parent's equity....
-
Explain which of these bonds has the absorption for its stretching vibration at higher wave number: (a) C H or C D (b) C = C or C C (c) C C1 or C I
-
Explain why the presence of a triple bond is much easier to detect in the IR spectrum of 1-hexyne than it is in the spectrum of3-hexyne. 80 60 O The sp-hybridized CH absorption bands: from 3000-2850...
-
Do you think the consequences SnapChat faced as part of its settlement with the FT are harsh enough to deter false claims? Explain.
-
Assume that P(A)=0.3, P(B)=0.4, P(AOBOC)=0.03, and P(AB)=0.88, then 1. the events A and B are, (A) Independent (B) Dependent (C) Disjoint (D) None of these. 2. P(CAB) is equal to, (A) 0.65 (B) 0.25...
-
A certain internet provider offers you an internet connection that provides download speeds up to 25 Mbps. How many users can access the system assuming each user is streaming video at a rate of 16...
-
On Aug 27, 2023, Sharmaine took out a $10,000 demand loan at 3.1% simple interest. On Oct 29, 2023, the interest rate changed to 2.3% simple interest and on December 31, 2023, the interest rate...
-
What protections do Green Credit, as the creditor, and its customers, as debtors, have in a credit relationship?
-
Aqueous sulfuric acid (HSO4) reacts with solid sodium hydroxide (NaOH) to produce aqueous sodium sulfate (NaSO4) and liquid water (HO). If 0.469 g of water is produced from the reaction of 2.9 g of...
-
In fitting a model to classify prospects as purchasers or nonpurchasers, a certain company drew the training data from internal data that include demographic and purchase information. Future data to...
-
(a) Prove that form an orthonormal basis for R3 for the usual dot product. (b) Find the coordinates of v = (1, 1, 1)T relative to this basis. (c) Verify formula (5.5) in this particular case. 48-65...
-
In 1990, the four-firm concentration ratio in the U.S. textbook industry was about 40 percent. The top four firms had an average of about 10 percent of the textbook market. The other 60 percent of...
-
Propose a mechanism to account for the formation of Bakelite from acid-catalyzed polymerization of phenol and formaldehyde.
-
Identify? the structural class to which the following polymer belongs, and show the structure of the monomer units used to make it:
-
Show the structures of the polymers that could be made from the following monomers (yellow-green =Cl): (b) (a)
-
Kaplan Inc. had 100,000 in unit-sales during the last quarter of last year. Managers expect the sales to increase by 10% each quarter over the next four quarters. The selling price is expected to...
-
This article titled "Evaluating Monetary Policy with Inflation Bands and Horizons 2023 -05 Troy Davig and Andrew Foerster February 21, 2023" discusses how monetary policy affects different sectors of...
-
Corporate governance is the set of rules, processes, and policies that guide and govern a business as it operates and manages risk. Tax laws have existed since the 1800s, and the history, Scope, and...

Study smarter with the SolutionInn App


