Question: For the ZrO 2 ?CaO system (Figure), write all eutectic and eutectoid reactions for cooling. Composition (mol% Cao) 10 20 30 40 50 3000 5000
For the ZrO2?CaO system (Figure), write all eutectic and eutectoid reactions for cooling.
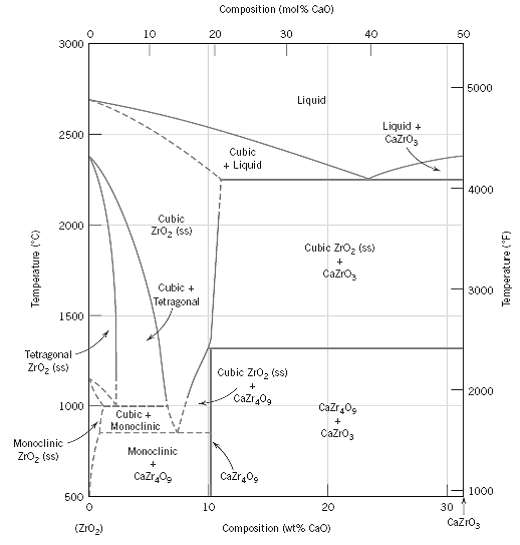
Composition (mol% Cao) 10 20 30 40 50 3000 5000 Liquid Liquid + Cazro, 2500 Cubic + Liquid 4000 Cubic 2000 Zro, (ss) Cubic Zro, (ss) Cazro, Cubic + 3000 Tetragonal 1500 Tetragonal Zroz (ss) Cubic ZIO, (ss) 2000 Cazr 0g 1000 Cazr,0g Cubic + Monoclinic Cazro3 Monoclinic Monoclinic ZrOz (ss) Cazr Og Cazr0g 1000 500 10 20 30 CazrO3 Composition (wt% CaO) (Z1O2) (0.) eameredwejL Temperature (F)
Step by Step Solution
3.42 Rating (168 Votes )
There are 3 Steps involved in it
There is only one eutectic for the portion of the ZrO 2 CaO system shown in ... View full answer

Get step-by-step solutions from verified subject matter experts
Document Format (1 attachment)
33-E-M-S-E-M-S (492).docx
120 KBs Word File


