Question: For thee compounds indicated whether the substituents are cis or trans whether they are axial or equatorial, whether the conformation shown or the other chair
For thee compounds indicated whether the substituents are cis or trans whether they are axial or equatorial, whether the conformation shown or the other chair conformation is more stable, and whether the compound shown or one of its stereo isomers is morestable.
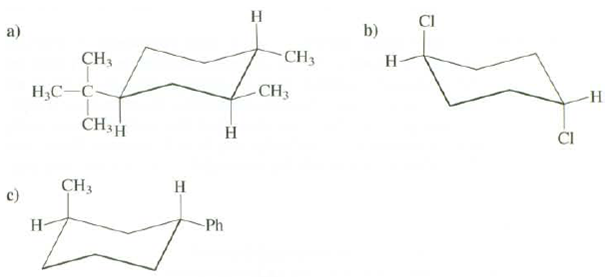
b) a) -CH3 CH3 H- ,- CH3 -H CH3H CI CH3 c) Ph
Step by Step Solution
3.47 Rating (160 Votes )
There are 3 Steps involved in it
In the representation of a cyclohexane chair conformation all bonds projecting closer to the top of ... View full answer

Get step-by-step solutions from verified subject matter experts
Document Format (1 attachment)
15-C-O-S (59).docx
120 KBs Word File


