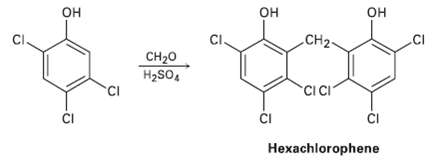
Hexachlorophene, a substance used in the manufacture of germicidal soaps, is prepared by reaction of 2, 4,
Question:
Hexachlorophene, a substance used in the manufacture of germicidal soaps, is prepared by reaction of 2, 4, 5-trichiorophenol with formaldehyde in the presence of concentrated sulfuric acid. Propose a mechanism for thereaction.
Transcribed Image Text:
н он он CI Он CI. CH2- сн20 H2SO, CIC CI ČI ČI Hexachlorophene
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (15 reviews)
DR1 Formaldehyde is protonated to form a carbocation 2 0 H CI OH H CHOH OH Base OH The formal...View the full answer

Answered By

Amit Kumar
I am a student at IIT Kanpur , which is one of the prestigious colleges in INDIA.
Cleared JEE Advance in 2017.I am a flexible teacher because I understand that all students learn in different ways and at different paces. When teaching, I make sure that every student has a grasp of the subject before moving on.
I will help student to get the basic understanding clear. I believe friendly behavior with student can help both the student and the teacher.
I love science and my students do the same.
4.90+
44+ Reviews
166+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The following reaction is important in the manufacture of sulfuric acid. At 900 K, 0.0216 mol of SO2 and 0.0148 mol of O2 are sealed in a 1.00-L reaction vessel. When equilibrium is reached, the...
-
A press produces parts used in the manufacture of large-screen plasma televisions. If the press is correctly adjusted, it produces parts with a scrap rate of 5%. If it is not adjusted correctly, it...
-
The strength of paper used in the manufacture of cardboard boxes (y) is related to the percentage of hardwood concentration in the original pulp (x). Under controlled conditions, a pilot plant...
-
R and Q form equal partnership RQ on February 15. Partner R has a tax year ending on July 31, whereas partner Qs tax year ends on October 31. What taxable year must the partnership use? a. February...
-
Determine the gross income of the beneficiaries in the following cases: a. Justin's employer was downsizing and offered employees an amount equal to one year's salary if the employee would...
-
A solid mixture weighing 0.5485 g contained only ferrous ammonium sulfate hexahydrate and ferrous chloride hexahydrate. The sample was dissolved in 1M H2SO4, oxidized to Fe3+ with H2O2, and...
-
To the left of z = 1.865 Find the indicated area under the standard normal curve. If convenient, use technology to find the area.
-
1. If you were advising Squires management team on their impending decision, what would you tell them? 2. Is there an alternative to reduce the impacts of high diesel prices other than to develop...
-
The Canoe Company... Net Income 2026 2025 2024-.- $11,146 $13,433 Flashy Life Vests 2026 ....2025.... 2024 $21,780 $20,710 Stockholders' Equity Common Stock 72,840 80,800 111,500 102,430 Retained...
-
Use the following selected accounts and amounts with normal balances from Juan Co. to prepare its classified balance sheet at December 31. Wages payable.. $ 1,000 Accounts receivable.. $ 7,000...
-
The compound MON-0585 is a nontoxic, biodegradable larvicide that is highly selective against mosquito larvae. Synthesize MON-0585 using either benzene or phenol as a source of the aromaticrings....
-
Benzenediazonium carboxylate decomposes when heated to yield N2, C02, and a reactive substance that can't be isolated. When Benzenediazonium carboxylate is heated in the presence of furan, the...
-
For each of the following incorrect entries, journalize the appropriate correcting entry(ies). a. The purchase of office supplies on credit for $1,800 was recorded as: b. A credit customer paid her...
-
What is a glycosidic linkage? What is the difference between alpha and beta glucose? Where do glyosidic linkages form? What is the difference between an alpha and a beta glyosidic linkage?
-
The Sweetwater Candy Company would like to buy a new machine that would automatically dip" chocolates. The dipping operation currently is done largely by hand. The machine the company is considering...
-
An art collector purchases a painting for $10,000. If the painting is expected to appreciate at an annual rate of 8%, what will be its estimated value in 10 years?
-
For the problem min, || Ax - bl, we have that the SVD of A is such that 1 2 1 0.5 0.3 If x has dimension 3 and A has full column rank, find the minimum error norm. A = USVT,UTb =
-
You are watching a production, one performer is overacting badly, to the point that he or she is quite unbelievable. Another performer is listless and has no energy. In each case, to what extent do...
-
Discuss two advantages of Hedge Fund B relative to Hedge Fund C with respect to investment characteristics. Sushil Wallace is the chief investment officer of a large pension fund. Wallace wants to...
-
On August 31, 2012, the balances of the accounts appearing in the ledger of Wood Interiors Company, a furniture wholesaler, are as follows:Prepare the August 31, 2012, closing entries for Wood...
-
N-butane is to be liquefied to make liquid petroleum gas (LPG). The butane is available at 25 C and 1 bar, it will be compressed to 15 bar in a compressor that has an isentropic efficiency of 85%,...
-
The oxymercuration reaction can be run in a methanol as the solvent rather than water. Predict the product of this reaction. 1) Hg(O,CCH)2, CH3OH 2) NaBH4, NAOH CH,CH,CH CH=CH;
-
The tautomerization of an enol to a ketone is catalyzed by either acid or base. In the acid-catalyzed mechanism H+ is added in the first step (see Figure). In the base-catalyzed mechanism, H+ is...
-
An unknown compound has the formula C6H10. (a) What is the DU for this compound? (b) When a solution of Br2 in CC14 is added to the unknown, the bromine color disappears. What information does this...
-
After you've completed this week's assigned reading, watch the video below. Next, respond to the Discussion questions underneath. Your first post should answer those questions and should be made by...
-
Factorize (x4 - 20x + 100)
-
Paragraph discussing food insecurity in the United States. What agencies in the federal government play an important role in ameliorating this situation? What groups of people are at particular risk?

Study smarter with the SolutionInn App


