The oxymercuration reaction can be run in a methanol as the solvent rather than water. Predict the
Question:
The oxymercuration reaction can be run in a methanol as the solvent rather than water. Predict the product of this reaction.
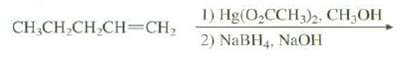
Transcribed Image Text:
1) Hg(O,CCH)2, CH3OH 2) NaBH4, NAOH CH,CH,CH CH=CH;
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (12 reviews)
In the oxymercuration reaction the mercury electrophile adds to the double bond ...View the full answer

Answered By

Anthony Ngatia
I have three academic degrees i.e bachelors degree in Education(English & Literature),bachelors degree in business administration(entrepreneurship option),and masters degree in business administration(strategic management) in addition to a diploma in business management.I have spent much of my life in the academia where I have taught at high school,middle level colleges level and at university level.I have been an active academic essays writer since 2011 where I have worked with some of the most reputable essay companies based in Europe and in the US.I have over the years perfected my academic writing skills as a result of tackling numerous different assignments.I do not plagiarize and I maintain competitive quality in all the assignments that I handle.I am driven by strong work ethics and a firm conviction that I should "Do Unto others as I would Like them to do to me".
4.80+
76+ Reviews
152+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
What kinds of methods can be run in a C# thread?
-
Methanol (CH3OH) can be made by the reaction of CO with H2: (a) Use thermochemical data in Appendix C to calculate (Ho for this reaction. (b) To maximize the equilibrium yield of methanol, would you...
-
The following reaction does not produce the product shown. (a) Predict the major product from the conditions shown above, and write a detailed mechanism for its formation. (b) What reaction...
-
In a LVDT, the two secondary voltages a. Are independent of the core position b. Are always in phase quadrature Oc. Vary equally depending on the core position O d. Vary unequally depending on the...
-
What is descriptive analytics? What are the various tools that are employed in descriptive analytics?
-
Highway curve with T = 265.00 ft, R = 1250 ft, and PI station = 87 + 33.55 ft. Tabulate R or D, T, L, E, M, PC, PT, deflection angles, and incremental chords to lay out the circular curves at full...
-
CD Real Estate specializes in residential real estate services in the state of California. To complement the experience and local market knowledge of its licensed realtors, CD Real Estate wants to...
-
At the end of February, Howard Productions accounting records reveal a balance for cash equal to $19,225. However, the balance of cash in the bank at the end of February is only $735. Howard is...
-
Workplace safety has economic, reputational, and ethical implications. What do you think these implications may be? what are the best aspects in employment law in the workplace?
-
A researcher wishes to compare the height (cm) of four plant varieties within a greenhouse. The greenhouse has six benches and each bench can accommodate 4 pots. The sunlight streaming in the...
-
Show the steps in the mechanism and predict the product that would be formed in thisreaction. Br2 CH,
-
The tautomerization of an enol to a ketone is catalyzed by either acid or base. In the acid-catalyzed mechanism H+ is added in the first step (see Figure). In the base-catalyzed mechanism, H+ is...
-
As we saw in Chapter 3, a group of MIT professors created a function for transforming student test scores of 0 to 100 into scores from 0 to 5, with transformed scores concentrated around 3, 4, and 5....
-
A 50% reaction (Parsons half-degree reaction) turbine is to be designed, having a mean diameter of 0.5 m and blade height of 30 mm. The blade angles are 60 degrees at inlet and 160 degrees at outlet....
-
A good writer anticipates the audience of a message. Although you cant always know exactly who the audience is, you can imagine the general characteristics of the receiver. Profiling your audience...
-
Suppose total reserve in bank A increased by $72,800,900. If required reserve ratio for the banking system is set at 5.9%, (0.059), a. Calculate bank A's required reserve. b. By how much will total...
-
The force and deflection reported in the table below, determine the observed spring constant k using the Least Squares Regression formula shown or Excel's =LINEST(). Report the result in N/mm with at...
-
After implementing the solution from the first question, Ben receives a complaint about users in his cafe hijacking other customers' web traffic, including using their usernames and passwords. How is...
-
Canadian Manufacturing Workers Workhours. The time plot in Figure 17.9 describes the average annual number of weekly hours spent by Canadian manufacturing workers (data are available in...
-
Find the intercepts and then graph the line. (a) 2x - 3y = 6 (b) 10 - 5x = 2y
-
At 35C, K = 1.6 10 -5 for the reaction If 2.0 moles of NO and 1.0 mole of Cl 2 are placed into a 1.0-L flask, calculate the equilibrium concentrations of all species. 2NOCI(g)2NO(g) + Cl(g)
-
Glucosamine, one of the eight essential monosaccharide?s (Section 25.7), is biosynthesized as its 6-phosphate derivative from fructose 6-phosphate by reaction with ammonia. Propose a mechanism....
-
Gentiobiose, a rare disaccharide found in saffron and gentian, is a reducing sugar and forms only n-glucose on hydrolysis with aqueous acid. Reaction of Gentiobiose with iodomethane and Ag20 yields...
-
Amygdalin, or laetrile, is a cyanogenic glycoside isolated in 1830 from almond and apricot seeds. Acidic hydrolysis of amygdalin liberates HCN, along with benzaldehyde and 2 equivalents of D-glucose....
-
1. Raman purchases a motor car from Bharathan whose cash price is Rs. 56,000 on 11.93. Rs. 15,000 is paid on signing the contract and the balance is to be paid in three equal annual instalments of...
-
Write a function that takes in a value x, a value el, and a list and adds as many el's to the end of the list as there are x's in the list. Make sure to modify the original list using list mutation...
-
Our office building has a total square footage of 120,000 square feet.We have 9 tenants in the building and no vacancies.The total square footage of the tenant spaces is 105,000.A) What is the Load...

Study smarter with the SolutionInn App


