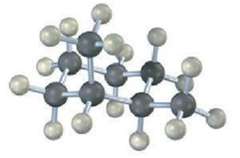
Question: How many electronically nonequivalent kinds of protons and how many kinds of carbons are present in the following compound? Don?t forget that cyclohexane rings can
How many electronically nonequivalent kinds of protons and how many kinds of carbons are present in the following compound? Don?t forget that cyclohexane rings can ring-flip.
Step by Step Solution
★★★★★
3.47 Rating (177 Votes )
There are 3 Steps involved in it
1 Expert Approved Answer
Step: 1 Unlock

If you assign R S configurations to the two carbons bonded to the methyl group it is ... View full answer

Question Has Been Solved by an Expert!
Get step-by-step solutions from verified subject matter experts
Step: 2 Unlock
Step: 3 Unlock
Document Format (1 attachment)
22-C-O-S (199).docx
120 KBs Word File


