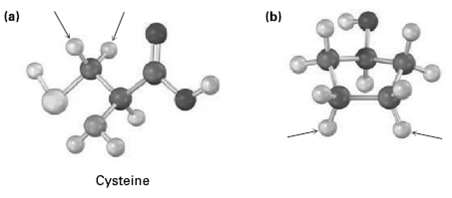
Identify the indicated protons in the following molecules as unrelated, homotopic, enantiotopic, ordiastereotopic: (a) (b) Cysteine
Question:
Identify the indicated protons in the following molecules as unrelated, homotopic, enantiotopic, ordiastereotopic:
Transcribed Image Text:
(a) (b) Cysteine
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (5 reviews)
a Because cysteine has a chirality center the indicated p...View the full answer

Answered By

Charles mwangi
I am a postgraduate in chemistry (Industrial chemistry with management),with writing experience for more than 3 years.I have specialized in content development,questions,term papers and assignments.Majoring in chemistry,information science,management,human resource management,accounting,business law,marketing,psychology,excl expert ,education and engineering.I have tutored in other different platforms where my DNA includes three key aspects i.e,quality papers,timely and free from any academic malpractices.I frequently engage clients in each and every step to ensure quality service delivery.This is to ensure sustainability of the tutoring aspects as well as the credibility of the platform.
4.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Identify the indicated hydrogen?s in the following molecules as pro-R or pro-S: (a) (b) .co2 H3N Alanine (S)-Glyceraldehyde
-
Identify the carbon atoms in the following molecules as primary, secondary, tertiary, orquaternary: H H CH CH CHCH2CHCH2CH Gs (c) (a) (b) CH2H2CH CHCCH2CCH CH
-
Label carbon in the following molecules as primary, secondary, tertiary, or quaternary.
-
The dehydration butanol of alumina is carried out over a silica-alumina catalyst at 680K. CH3CH2CH2CH20H------->cat CH3CH=CHCH3 + H2O The rate law is -r Bu = KPBU/(1+KBuPBul with k= 0.054...
-
Is the following statement correct? "Double marginalization helps firms because it enables them to raise prices."
-
A researcher wishes to see if there is a difference between the mean number of hours per week that a family with no children participates in recreational activities and a family with children...
-
National Life Expectancies. We continue the analysis begun in Exercises 1.7and 2.22 Now fit a regression model on LIFEEXP using three explananatural logarithmic transform of PRIVATEHEALTH). a....
-
On January 1, the company issued 10-year bonds with a face value of $200,000. The bonds carry a coupon rate of 10%, and interest is paid semiannually. On the issue date, the market interest rate for...
-
Schedule of Cost of Goods Manufactured Sydney Company reported the following amounts for October: Beginning raw materials inventory $144,000 Beginning work-in-process inventory 252,000 Beginning...
-
SummerFun, Inc., produces a variety of recreation and leisure products. The production manager has developed an aggregate forecast: Use the following information to develop aggregate plans. Develop...
-
How many electronically nonequivalent kinds of protons and how many kinds of carbons are present in the following compound? Don?t forget that cyclohexane rings can ring-flip.
-
The following 1H NMR absorptions were obtained on a spectrometer operating at 200MHz and are given in hertz downfield from the TMS standard. Convert the absorptions to units. (a) 436 Hz (b) 956 Hz...
-
Which of the statements listed is true regarding the following redox reaction occurring in a spontaneous electrochemical cell? Mg(s) + Cu 2+ (aq) Cu(s) + Mg 2+ (aq) (a) Mg is oxidized at the anode....
-
DIET PROBLEM. Grant Winfield is a 71-year-old grandfather who likes to mix breakfast cereals together for taste and as a means of getting at least 50% of the recommended daily allowances (RDA) of...
-
Compare and contrast zero-based versus incremental budgeting? Zero-based budgets (ZBB) start with just that, zeroed out or a clean slate. In this type of budget, each and every item needs to be...
-
Louise has been saving to pay for her daughter June's college education. June just turned 10 at (t=0), and she will be entering college 8 years from now (at t = 8). College tuition and expenses at...
-
How do nuclear bodies, such as Cajal bodies, PML bodies, and speckles, serve as dynamic subnuclear structures involved in RNA metabolism, transcriptional regulation, and genome maintenance, and how...
-
What are the linkages among financial decisions, return, risk and stock value for this firm? The firm I selected for this discussion is Nike. Nike is the #1 brand in sporting goods industry, regarded...
-
Suppose you are given a random variable that has the standard normal distribution. a. Find the value of \(z\) such that \(5 \%\) of all values of this random variable will be greater. b. Find the...
-
QUESTION 2 The CEO of Farisha Hijab Sdn Bhd insisted on further investigation to be carried out that he also required Mr Muaz to conduct the analysis of variance for the material and labour of the...
-
Calculate the vapor pressure of a solution containing 24.5 g of glycerin (C 3 H 8 O 3 ) in 135 mL of water at 30.0 C. The vapor pressure of pure water at this temperature is 31.8 torr. Assume that...
-
Draw the expected broadband-decoupled 13C NMR spectra of the following compounds. Use Figure 13-41 (page 603) to estimate the chemical shifts. (a) (b) (c) (d) C CH H,C CH C-H C-C
-
(a) Show which carbon atoms correspond with which peaks in the 13C NMR spectrum of butan-2-one (Figure 13-45). (b) Draw the proton NMR spectrum you would expect for butan-2-one. How well do the...
-
Repeat Problem 13-25, sketching the off-resonance-decoupled 13C spectra of the compounds. Problem 13-25 Draw the expected broadband-decoupled 13C NMR spectra of the following compounds. Use Figure...
-
Draw the shear diagram for the beam. Set P = 600 lb, a = 5 ft, b = 7 ft. Draw the moment diagram for the beam.
-
Ahmed, Bassem and Jamal are partners in Tadamon Company who share profits and losses at a ratio of 2 : 2 : 4 respectively. On 1 / 1 / 2 0 2 2 , it was decided to liquidate the company, and the...
-
Draw the shear diagram for the beam. Follow the sign convention. Draw the moment diagram for the beam. Follow the sign convention.

Study smarter with the SolutionInn App


