Occasionally, chemists need to invert the stereochemistry of an alkene?that is, to convert a cis alkene to
Question:
Occasionally, chemists need to invert the stereochemistry of an alkene?that is, to convert a cis alkene to a trans alkene, or vice versa. There is no one-step method for doing an alkene inversion, but the transformation can be carried out by combining several reactions in the proper sequence. How would you carry out the following reactions?
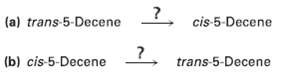
Transcribed Image Text:
5 cis-5-Decene (a) trans 5-Decene (b) cis-5-Decene trans-5-Decene
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 72% (11 reviews)
a b trans5Decene cis5Decene ...View the full answer

Answered By

Rohail Amjad
Experienced Finance Guru have a full grip on various sectors, i.e Media, Insurance, Automobile, Rice and other Financial Services.
Have also served in Business Development Department as a Data Anlayst
4.70+
32+ Reviews
83+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
The production of 1,3-butadiene can be carried out by the dehydrogenation of 1- butene: C2H5CH:CH2(g) CH2:CHCH:CH2(g) + H2(g) Side reactions are suppressed by the introduction of steam. If...
-
The production of 1,3-butadiene can be carried out by the dehydrogenation of n- butane: C4H 10(g) CH2 : CHCH : CH2(g) + 2H2(g) Side reactions are suppressed by the introduction of steam. If...
-
How would you carry out the following reactions to introduce deuterium into organicmolecules? (a) CHH2C3CCH2C Hs 2H5 (b) C2H5 CHH2C3CCH2CH3 C=C Hs D (c) CH3CH2CH2C=CH CH3CH2CH2C=CD CD CD2 C=CH (d)
-
Super Slushie charges $6.50 for a medium slushie and $9 for a large slushie. Their total net marketing contribution is $28,000 per week. They want to raise the price of each slushie 12.5% next month...
-
You are organizing rides for a group of campers going on an all-day, off-site trip. You have lined up some drivers, and your problem is to assign campers to drivers. The drivers and the capacities of...
-
An electric pencil sharpener rated 240 mW, 6 V is connected to a 9-V battery as shown in Fig. 2.138. Calculate the value of the series-dropping resistor Rx needed to power the sharpener. Figure 138...
-
Platinum Status Ltd uses an annual flexible budget based on standard direct machine hours for the following factory overhead items. Required (a) Why is a flexible budget performance report a better...
-
The City Commission of Nashville has decided to build a botanical garden and picnic area in the heart of the city for the recreation of its citizens. The precedence table for all the activities...
-
The Computing department of the University would like to implement its catalogue of computing devices as a doubly linked list, called DeviceList. 1. Write a Device node node class, called DeviceNode,...
-
Do you think you will ever have an extreme job? Are you sure? Explain. Why do you think the number of extreme jobs has risen? Do you think organizations should encourage extreme jobs, discourage...
-
How would you carry out the followingreactions? (a) CCH-CCH3 CH3CH2C=CH () H2C%3CH CH3CH2CH2CHO .3- (c) 3C (d) CH . CH 7, 22H (e) CH3CH2C=CH (f) CH3CH2CH2CH2CH=CH2 CHCH2CH2CH2C%CH (2 steps)
-
Propose structures for hydrocarbons that give the following products on oxidative cleavage by KMnO4 orO3: (b) .Co2 (a) O2 + CH(CH2)5C02H CC2H + e) O2CICH2)8CO2H (d) CH + 2H2cO2H + co2 (e) CO2...
-
What is the transverse stiffness of a cantilever steel beam \(\left(\mathrm{E}=200 \times 10^{9} \mathrm{~N} / \mathrm{m}^{2} ight)\) with a length of \(10 \mu \mathrm{m}\) and a rectangular cross...
-
Who is a great example of a Transactional Leadership and Laissez-Faire Leadership in the 20 century?
-
How might the consequences of the situation have been different if acted as a Creator?
-
About costs incurred after the acquisition, what does increased service potential mean?
-
Define the significant differences and similarities between empowerment and laissez-faire leadership.?
-
If you were the leader of your organization, how would you change the way your organization communicates in order to assure that all employees contribute to the organization's mission, vision, and...
-
A: When a month is randomly selected and ripped from a calendar and destroyed, it is July. B: When a different month is randomly selected and ripped from a calendar, it is November. Independent and...
-
The May 2014 revenue and cost information for Houston Outfitters, Inc. follow: Sales Revenue (at standard).............. $ 540,000 Cost of Goods Sold (at standard) ..........341,000 Direct Materials...
-
To what volume should you dilute 50.0 mL of a 12 M stock HNO 3 solution to obtain a 0.100 M HNO 3 solution?
-
Draw Lewis structures for (a) Two compounds of formula C4H10 (b) Two compounds of formula C2H6O (c) Two compounds of formula C2H7N (d) Three compounds of formula C2H7NO (e) Three compounds of formula...
-
Draw a complete structural formula and a condensed structural formula for (a) Three compounds of formula C3H8O (b) Five compounds of formula C3H6O
-
Write Lewis structures for the following molecular formulas. (a) N2 (b) HCN (c) HONO (d) CO2 (e) CH3CHNH (f) HCO2H (g) C2H3CI (h) HNNH (i) C3H6 (one double bond) (j) C3H4 (two double bonds) (k) C3H4...
-
Zach Ltd. produced the following data for the year 2015 - o Interest Income $4,650,000 o Capital Gain realized $50,000 o Dividend Income from company's 15% ownership in XYZ Ltd. $65,000 o Corporate...
-
A project requires an increase in net working capital of $150,000 at time 0 that will be recovered at the end of its 10 year life. If the opportunity cost of capital is 8%, what is the effect of NWC...
-
A firm is 40% financed by debt with a yield-to-maturity of 8.5%. The equity has a beta of 1.3, the market risk premium is 8.4% and the risk-free rate is 3.8%. What is the firm's WACC if the tax rate...

Study smarter with the SolutionInn App


