How would you carry out the following transformation, a step used in the commercial synthesis of(S)-ibuprofen? CN
Question:
How would you carry out the following transformation, a step used in the commercial synthesis of(S)-ibuprofen?
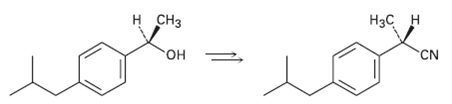
Transcribed Image Text:
н снз Нас н он CN
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (15 reviews)
Strategy Recall from Chapter 11 that OH is a very poor leaving group in re...View the full answer

Answered By

Kalyan M. Ranwa
I have more than seven years of teaching experience in physics and mechanical engineering.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
How would you carry out the following synthesis (more than one step is required)? What stereo chemical relationship between the ?CO 2 CH 3 group attached to the cyclohexane ring and the ?CHO groups...
-
How would you carry out the following transformations? More than one step may berequired. H-CH (a) (b) Br - (c) " - " - (d) CH3CH2CH2CH2C=CH CHH2CH2CH2CH2H20CH3 H (e) CH3CH2CH2CH2CHCH3...
-
How would you carry out the following transformations? Co .CO2H (a) (b) Co CH2 C (c) CH2SH
-
Jeremy acquired the following ordinary shares in Scarlon plc: He made no further acquisitions during 2021. On 22 December 2020, he sold 10,000 shares in the company for 10 per share. Calculate the...
-
How do functional tactics differ from corporate and business strategies?
-
How large a sample would be needed to ensure that the length of the CI in Problem 6.30 is 0.5 g/mL assuming the sample standard deviation remains at 2.0 g/mL? Pharmacology Suppose we want to estimate...
-
The dataset NFLContracts 2015 contains the yearly salary (in millions of dollars) from the contracts of all players on a National Football League (NFL) roster at the start of the 2015 season. (a) Use...
-
Solve the linear programming model formulation in Problem 41 for Island Publishing Company graphically. a. Determine the sensitivity range for the advertising revenue generated by the real estate...
-
A 2.2-kg fire rocket travels east 50 meters in 4.2 seconds and then it explodes and breaks apart into 3 pieces. The first piece is 1.2-kg and travels west at 7 m/s. The second piece is 0.7-kg and...
-
L & M Power In the next two years, a large municipal gas company must begin constructing new gas storage facilities to accommodate the Federal Energy Regulatory Commissions Order 636 deregulating the...
-
Use the reaction of a Grignard reagent with a carbonyl compound to synthesize the following compound:
-
What product(s) would you expect from dehydration of the following alcohols with POCl3 in pyridine? Indicate the major product in eachcase? (a) (b) (c) CH3CH2CHCHCH3 CH CH3 H (d) (e) CCCH2CH...
-
Express each radical in simplest form, rationalize denominators, and perform the indicated operations. 32a6b4+3a/243ab
-
Valley Bank has an average interest rate of 10% on %500 million of loans and securities, an average interest rate of 5% on $400 million of deposits and other borrowed funds, noninterest revenue of...
-
A shop has implemented a new coupon discount program for its customers. Employees could be scanning coupons but charging the customers full price and then pocketing the difference.) explain,construe...
-
Discuss the relationship of confirmation procedures to the auditor's assessment of audit risk. Discuss certain factors that affect the reliability of confirmations
-
An all-equity firm has a beta of 1.03. The firm is evaluating a project that will increase the output of the firm's existing products. The market risk premium is 6.9 percent, and the risk-free rate...
-
From the investor's perspective, briefly describe the cash flows associated with a bond. Briefly explain the term yield to maturity.
-
Do you think a trial judge should be allowed to summarize the evidence for the jurys benefit before the jurors retire to deliberate? What advantages and disadvantages can you see in such a practice?...
-
Would you expect an ionic solid or a network covalent solid to have the higher melting point?
-
First, complete and balance each of the equations below. Then, choosing among ethanol, hexane, and liquid ammonia, state which (there may be more than one) might be suitable solvents for each of...
-
Dimethylformamide (DMF), HCON(CH3)2, is an example of a polar aprotic solvent, aprotic meaning it has no hydrogen atoms attached to highly electronegative atoms. (a) Draw its dash structural formula,...
-
As noted in Table 3.1, the pKa of acetone, CH3COCH3, is 19.2. In Table 3.1 (a) Draw the bond-line formula of acetone and of any other contributing resonance form. (b) Predict and draw the structure...
-
I am running for Vice President of Legislative Affairs at my University as part of the Student Government Association. I am need of ideas!!! Keep this in mind. My platform is centered around pushing...
-
What are some ways that synergistic communication can positively affect organizational communication? Furthermore, how does SC relate to the achieving of (or the not achieving of) the overall goals...
-
Choose an organization's website. Provide the name of the company and the URL address at the top of your answer. i) Analyze the website using THREE (3) relevant corporate communication concepts. (9%)...

Study smarter with the SolutionInn App


