How would you carry out the following reactions to introduce deuterium into organicmolecules? (a) CHH2C3CCH2C Hs 2H5
Question:
How would you carry out the following reactions to introduce deuterium into organicmolecules?
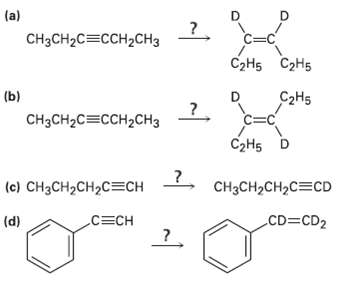
Transcribed Image Text:
(a) CHзсH2C3CCH2CНз СэHs С2H5 (b) C2H5 CHзсH2C3CCH2CH3 C=C СэHs D (c) CH3CH2CH2C=CH CH3CH2CH2C=CD CD CD2 C=CH (d)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 56% (16 reviews)
a b c d CH3CHCCCHCH3 CH3CHCCCHCH3 CH3CH...View the full answer

Answered By

Vikash Gupta
I am graduated in Physics in 2018, from KIRORIMAL COLLEGE, University of Delhi. Now I am persuing Master's degree in physics. I like to do physics problems. I have experience of 1 year in tutoring. I think Physics is the only subject where you understand things,how they are happening . In physics you learn Maths and apply it. So I would like to join your platform to solve many Physics problems.
5.00+
5+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
How would you carry out the following transformations? More than one step may berequired. H-CH (a) (b) Br - (c) " - " - (d) CH3CH2CH2CH2C=CH CHH2CH2CH2CH2H20CH3 H (e) CH3CH2CH2CH2CHCH3...
-
How would you carry out the following transformation, a step used in the commercial synthesis of(S)-ibuprofen? CN
-
How would you carry out the following transformations? Co .CO2H (a) (b) Co CH2 C (c) CH2SH
-
YOU have just graduated from Deakin as an accounting and finance graduate. During your time at Deakin, you were organized and diligent with your studies and thus graduated with flying colors. This...
-
A manufacturer of nylon carpets produces rolls of carpeting at four factories and ships them to distributors in five locations The following table shows the capacities at the factories and the...
-
Calculate v1 and v2 in the circuit of Fig. 3.62 using nodal analysis. Figure 3.62 AW 8 0 4 3 A
-
Eccentric Electric Ltd operates three selling departments mountain bike, road bike, and hybrid. Certain indirect expenses are allocated to the selling departments as follows. Required Prepare a...
-
March 31, 2014, adjusted trial balance for Brenner Climbing Adventures has been alphabetized as follows: Required Journalize the closing entries. No. Account Debit Credit $ 2,600 168 Accumulated...
-
The ability of technology to diagnosis diseases earlier and provide treatments certainly has had an impact on people living longer. Many health care dollars are spent on treatments such a dialysis,...
-
Requirements: Theme :future of biotechnology Read a recent news article or a blog related to the theme. Write Introduction (200 words) Summary and theoretical application related to course (400...
-
How would you synthesize the following compounds from acetylene and any alkyl halides with four or fewer carbons? More than one step may herequired. (a) CH3CH2CH2C=CH (b) H2C%3CCH CH CH CHH2H3D (d)...
-
How would you prepare Cyclodecyne starting from acetylene and any alkyl halide needed?
-
Jason Co. accepts VISA and MasterCard for any sales transaction exceeding $50.The company has not yet implemented online recording of the credit card transaction, but does have a toll-free number to...
-
What areas if any does US health care system outrank other countries? Do you feel that these strengths helps to validate the rising costs of health care ? Why or why not
-
Find a formula for the inverse of the following function, if possible. 5 W(x) 2x-2
-
What might be the impact of laissez faire leadership style on health care safety and quality outcomes?
-
Consider the following scenario: You are the safety manager for a team that is working to 10-story addition to a hospital. The framework of the addition will be steel and concrete. The floors will be...
-
7) The XYZ Company has estimated expected cash flows for 1996 to be as follows: Probability Cash flow .10 $120,000 .15 140,000 .50 150,000 .15 180,000 .10 210,000 Calculate: a. expected value b....
-
A: When an M&M is randomly selected from the 100 M&Ms listed in Data Set 20, it is one of the 8 yellow M&Ms. B: When a second different M&M is randomly selected from those listed in Data Set 20, it...
-
Repeat Exercise 16.6 using the t-test of the coefficient of correlation. Is this result identical to the one you produced in Exercise 16.6?
-
What is the molarity of Cl in each solution? a. 0.200 M NaCl b. 0.150 M SrCl 2 c. 0.100 M AlCl 3
-
The following compounds are listed in increasing order of acidity. In each case, the most acidic proton is shown in red. (a) Show the structure of the conjugate base of each acid, including any...
-
Amides such as acetamide are much weaker bases than amines, such as ethylamine (CH3CH2NH2). (a) Use resonance forms to show why the nonbonding electrons on the nitrogen atom of the amide are very...
-
Methyllithium (CH3Li) is often used as a base in organic reactions. (a) Predict the products of the following acid-base reaction. CH3CH2 - OH + CH3 - Li (b) What is the conjugate acid of CH3Li?...
-
The hawkins company maintains one set of financial records for financial reporting purpose. separate records are also kept for tax compliance purpose. why is that necessary?
-
Managerial accounting must follow GAAP. Financial accounting does not. There is no significant difference between financial and managerial accounting. Managerial accounting provides information for...
-
Troy Abraham had the following assets and liabilities as of October 1, 2019. Assets Liabilities Cash $6,500 Automobile $8,700House $165,000 Contents of Home $6,400 Prepaid Insurance $1,700 Unpaid...

Study smarter with the SolutionInn App


