How would you carry out the following synthesis (more than one step is required)? What stereo chemical
Question:
How would you carry out the following synthesis (more than one step is required)? What stereo chemical relationship between the ?CO2CH3 group attached to the cyclohexane ring and the ?CHO groups would your synthesis produce?
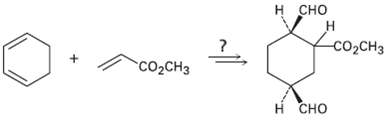
Transcribed Image Text:
н сно H. со,CHз "сооснз н сно
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 61% (13 reviews)
Although it is usually best to work backwards in a synthesis ...View the full answer

Answered By

Susan Juma
I'm available and reachable 24/7. I have high experience in helping students with their assignments, proposals, and dissertations. Most importantly, I'm a professional accountant and I can handle all kinds of accounting and finance problems.
4.40+
15+ Reviews
45+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
How would you carry out the following transformations? Co .CO2H (a) (b) Co CH2 C (c) CH2SH
-
How would you carry out the following transformations? "Co CH2 C Lc
-
How would you carry out the followingreactions? (a) CCH-CCH3 CH3CH2C=CH () H2C%3CH CH3CH2CH2CHO .3- (c) 3C (d) CH . CH 7, 22H (e) CH3CH2C=CH (f) CH3CH2CH2CH2CH=CH2 CHCH2CH2CH2C%CH (2 steps)
-
in an armicie entitled Fuel Economy Calculations tio Be Alteted lames Healey indicated that the goermnt planned to change how it caleulates fuel economy for new cas and trucks Thia in the first...
-
How could the services firm in the second example have met the requirements for overtime pay?
-
Metallic magnesium can be made by the electrolysis of molten MgCl2. (a) What mass of Mg is formed by passing a current of 4.55 A through molten MgCl2, for 4.50 days? (b) How many minutes are needed...
-
Which of the following procedures is most telling in addressing the existence assertion for accounts payable? a. Test for unrecorded liabilities. b. Confirm balances with creditors. c. Verify the...
-
The trial balances before and after adjustment for Frinzi Company at the end of its fiscal year are presented below. Instructions Prepare the adjusting entries that weremade. FRINZI COMPANY Trial...
-
Determine the size of a septic 10 points tank to serve 12 persons for small residential house. Assume 0.9m width and 1.2m depth of the tank. Your answer A hotel with private baths facilities will...
-
Part 3 - Coding Exercise: Decoding a Message from a Text File In this exercise, you will develop a function named `decode(message_file)`. This function should read an encoded message from a .txt file...
-
Dimethyl butynedioate also undergoes a DielsAlder reaction with (2E, 4Z)-hexadiene, but the stereochemistry of the product is different from that of the (2E, 4E) isomer (Problem 14.55). Explain.
-
The double bond of an enamine (alkene amine) is much more nucleophilic than a typical alkene double bond. Assuming that the nitrogen atom in an enamine is sp2-hybridized, draw an orbital picture of...
-
Is there sufficient evidence to infer that Americans born in the United States and those born elsewhere have different incomes (INCOME)?
-
Nittany Company uses a periodic inventory system. At the end of the annual accounting period, December 31 of the current year, the accounting records provided the following information for product 1:...
-
Harris Fabrics computes its plantwide predetermined overhead rate annually on the basis of direct labor-hours. At the beginning of the year, it estimated that 29,000 direct labor-hours would be...
-
Given the following two points of an exponential function, determine the equation of the exponential function. (-3,81), (0,3)
-
How can resource management be effectively implemented in agile and dynamic project environments, where flexibility and adaptability are paramount?
-
A stock is currently priced at $154. If the dividend that was just paid (D0) was $8.96, and dividends were expected to grow at a constant rate of 1.7%, what is the required return on the stock, rs?
-
The Tesla Roadster, a production electric car, has a \(375 \mathrm{~V}\) battery system that can provide a power of \(200 \mathrm{~kW}\). At this peak power, what is the current supplied by the...
-
Frontland Advertising creates, plans, and handles advertising campaigns in a three-state area. Recently, Frontland had to replace an inexperienced office worker in charge of bookkeeping because of...
-
What volume (in mL) of a soft drink that is 10.5% sucrose (C 12 H 22 O 11 ) by mass contains 78.5 g of sucrose? (The density of the solution is 1.04 g/mL.) SORT You are given a mass of sucrose and...
-
Predict the major products formed when the following amines undergo exhaustive methylation, treatment with Ag2O, and heating. (a) Hexan-2-amine (b) 2-methylpiperidine (c) N-ethylpiperidine (d) (e)...
-
Give the products expected when the following tertiary amines are treated with a peroxyacid and heated. (a) N, N-dimethylhexan-2-amine (b) N, N-diethylhexan-2-amine (c) Cyclohexyldimethylamine (d)...
-
When the (R, R) isomer of the amine shown is treated with an excess of methyl iodide, then silver oxide, then heated, the major product is the Hofmann product. (a) Draw the structure of the major...
-
Find the NPV of the project below if your company WACC = 10% and the project has comparable risk to your firm's average risk. The project requires an immediate investment of $50,000 and generates a...
-
Performance appraisals are used throughout business to provide feedback and growth opportunities to employees; this in turn leads to increased productivity. Leaders create performance appraisals for...
-
Question 3 (4 marks) Students at the school who are studying mathematics in Years 7 to 10 can receive one of three grades at the end of the year: distinction (D), credit (C) or pass (P). A regular...

Study smarter with the SolutionInn App


