Question: How would you carry out the following transformations using an organo copper coupling reaction? More than one step is required in eachcase. (a) CH (b)
How would you carry out the following transformations using an organo copper coupling reaction? More than one step is required in eachcase.
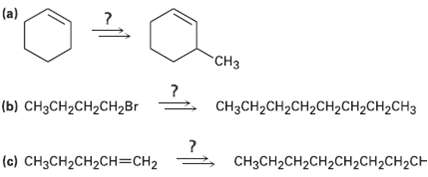
(a) "CH (b) HH2CH2CHBr CH3CH2CH2CH2CH2CH2CH2CH3 (c) CH3CH2CH2CH=CH2 CHCH2CH2CH2CH2H2CH2CH
Step by Step Solution
3.39 Rating (171 Votes )
There are 3 Steps involved in it
a Strategy The methyl group has an allylic relationship to the double bond Thus an organometallic co... View full answer

Get step-by-step solutions from verified subject matter experts
Document Format (1 attachment)
22-C-O-O-H (11).docx
120 KBs Word File


