How would you carry out the following transformations? Tell the reagents you would use in each case.
Question:
How would you carry out the following transformations? Tell the reagents you would use in each case.
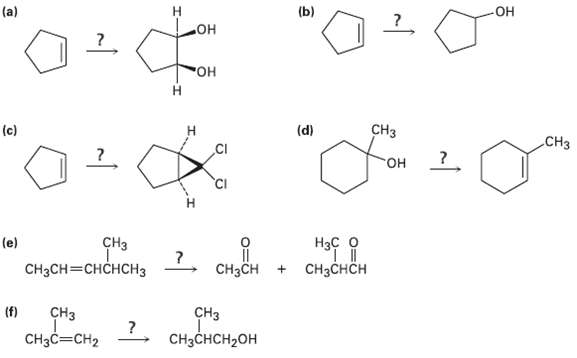
Transcribed Image Text:
(a) (b) Он но° "Он н CHз (c) (d) CHз но- CHз CH3CH=CHCHCH3 (e) Нас о ?. CнзCH + CHзснсH (f) CHз CHз CHзсHCH-оH CH3C=CH2
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 90% (10 reviews)
a b OsO4 NMO H I OH OH OH 1 HgOAc2 H2O 2 NaBH4 Hy...View the full answer

Answered By

Brown Arianne
Detail-oriented professional tutor with a solid 10 years of experience instilling confidence in high school and college students. Dedicated to empowering all students with constructive feedback and practical test-taking strategies. Effective educator and team player whether working in a school, university, or private provider setting. Active listener committed to helping students overcome academic challenges to reach personal goals.
4.60+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
How would you carry out the following transformations using an organo copper coupling reaction? More than one step is required in eachcase. (a) "CH (b) HH2CH2CHBr CH3CH2CH2CH2CH2CH2CH2CH3 (c)...
-
How would you carry out the following transformations? More than one step may berequired. H-CH (a) (b) Br - (c) " - " - (d) CH3CH2CH2CH2C=CH CHH2CH2CH2CH2H20CH3 H (e) CH3CH2CH2CH2CHCH3...
-
How would you carry out the following transformation, a step used in the commercial synthesis of(S)-ibuprofen? CN
-
Show that the angular wave number k for a non-relativistic free particle of mass m can be written as in which K is the particle's kineticenergy 27 V2mK k =
-
Calculate each Poisson probability: a. P(X = 6), = 4.0 b. P(X = 10), = 12.0 c. P(X = 4), = 7.0
-
The 304-stainless-steel cylinder has an inner diameter of 4 in. and a wall thickness of 0.1 in. If it is subjected to an internal pressure of p = 80 psi, axial load of 500 lb, and a torque of 70 lb...
-
In each of the following situations, indicate the statistical analysis you would conduct and the appropriate test or test statistic that should be used. a. Consumer preferences for Coca-Cola were...
-
Heller Manufacturing has two production facilities that manufacture baseball gloves. Production costs at the two facilities differ because of varying labor rates, local property taxes, type of...
-
Calculate the solution set of the following system, using Gauss-Jordan elimination method (that is by transforming the augmented matrix of the system in its reduced row echelon form) (a) x+y+z+w= 5...
-
Production engineers at Sinotron believe that a modified layout on its assembly lines might increase average worker productivity (measured in the number of units produced per hour). However, before...
-
Predict the products of the following reactions, showing both regiochemistry and stereochemistry where appropriate: CH (b) (a) 1. 2. Zn, H* M H* CH 1. Hg(OAc)2, H20 2. NABH4 (c) (d) C 1. 2. Hz, "
-
Which reaction would you expect to be faster, addition of HBr to cyclohexene or to 1-methylcyclohexane? Explain.
-
In Exercises 1558, find each product. (7x 3 + 5)(x 2 - 2)
-
Many companies locate manufacturing activities in countries with low labour costs and few labour laws. What ethical obligations do businesses have when operating in such countries?
-
With reference to case law, critically discuss the constitutionality of section 23(1)(d) and section 189(1) of the Labour Relations Act (South Africa) NELSON MANDELA UNIVERSITY Question 2...
-
discuss the major political ideologies in the united sates and their impact on international human resource management in relation to human resource functions, strategic business relations and labour...
-
What policies could be placed to curb the negative influences of technology on the labor market?
-
https://www.youtube.com/watch?v=wRNiPSenS_M&ab_channel=TheMacquarieUniversityCommunity Advise us on the labour laws and industrial relations system in France please.
-
Determine the equation of the phase plane trajectory of the differential equation \[\frac{d^{2} x}{d t^{2}}+\frac{d x}{d t}=0\] with the initial conditions \(x(0)=0\) and \(\dot{x}(0)=1\).
-
Beginning with a country that has a trade deficit, demonstrate graphically what will happen to a countrys potential output with globalization if that countrys costs of production fall. Explain your...
-
Methanol has a normal boiling point of 64.6 C and a heat of vaporization (H vap ) of 35.2 kJ/mol. What is the vapor pressure of methanol at 12.0 C? SORT You are given the normal boiling point of...
-
Predict the products, including their stereochemistry, from the E2 reactions of the following diastereomers of stilbene dibromide with sodium ethoxide in ethanol. Assume that one equivalent of HBr is...
-
Draw the structure of the starting material that would undergo azir-elimination give the -E isomer of the alkene product in the E2 reaction of Eq. 9.40.
-
Draw the structure of the starting material that would undergo azir-elimination give the -E isomer of the alkene product in the E2 reaction of Eq. 9.40.
-
A taxpayer disposed a part of asset for 100,000 SR in cash. The taxpayer purchased that asset for 90,000 SR 5 years ago but the market value of the complete assets was as follows: Part disposed...
-
A company maintains debt/equity ratio of 0.6. The flotation cost of equity is 12% and flotation cost of debt it is 6%. The firm is considering a new project which will require $5 million in external...
-
Flight Caf prepares in-flight meals for airlines in its kitchen located next to a local airport. The company's planning budget for July appears below: Flight Caf Planning Budget For the Month Ended...

Study smarter with the SolutionInn App


