In Figure particle 1 (of charge + 1.00?C), particle 2 (of charge + 1.00?C), and particle 3
Question:
In Figure particle 1 (of charge + 1.00?C), particle 2 (of charge + 1.00?C), and particle 3 (of charge Q) form an equilateral triangle of edge length a. For what value of Q (both sign and magnitude) does the net electric field produced by the particles at the center of the triangle vanish?
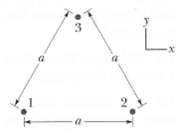
Transcribed Image Text:
3 10
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 55% (9 reviews)
On the one hand the conclusion that Q 100 C is clear fr...View the full answer

Answered By

Jonas Araujo
I have recently received the degree of PhD. In Physics by the Universidade Federal do Maranhão after spending a term in Durham University, as I have been awarded a scholarship from a Brazilian mobility program. During my PhD. I have performed research mainly in Theoretical Physics and published works in distinguished Journals (check my ORCID: https://orcid.org/0000-0002-4324-1184).
During my BSc. I have been awarded a scholarship to study for a year in the University of Evansville, where I have worked in detection-analysis of photon correlations in the the Photonics Laboratory. There I was a tutor in Electromagnetism, Classical Mechanics and Calculus for most of that year (2012).
I am very dedicated, honest and a fast learner, but most of all, I value a job well done.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Fundamentals of Physics
ISBN: 978-0471758013
8th Extended edition
Authors: Jearl Walker, Halliday Resnick
Question Posted:
Students also viewed these Electricity and Magnetism questions
-
In Figure particle 1 of charge + 4e is above a floor by distance d1 = 2.00 mm and particle 2 of charge + 6e is on the floor, at distance d2 = 6.00 mm horizontally from particle 1. What is the -r...
-
In Figure particle 1 of charge q1 = + e and particle 2 of charge q2 = 5e are fixed on an x axis. Distance d = 5.60pm. What is the electric potential difference VA VB?
-
In Figure particle 1 of charge ?? 80.0C and particle 2 of charge + 40.0C are held at separation L = 20.0 cm on an x axis. In unit-vector notation, what is the net electrostatic force on particle 3,...
-
Pettit Printing Company has a total market value of $100 million, consisting of 1 million shares selling for $50 per share and $50 million of 10 percent perpetual bonds now selling at par. The...
-
Review your results from Short Exercise SB-10. Requirements 1. Total each column of the cash payments journal. 2. Open the following four-column accounts in the accounts payable subsidiary ledger:...
-
An investor is looking for arbitrage opportunities in the Treasury bond futures market. What complications are created by the fact that the party with a short position can choose to deliver any bond...
-
Design a heat exchanger network for MER with at most 15 heat exchangers (including utility heaters) and \(\Delta T_{\text {min }}=10^{\circ} \mathrm{C}\) for the following streams: When MER targets...
-
1. What categories of costs would you expect to see in a list of MooBella start-up costs? 2. What was the mix of fundsby percentageused by MooBella to get started? 3. What are the start-up costs that...
-
Martinez Company's ending inventory includes the following items. Product Helmets Units Cost per Unit Market per Unit 33 $ 55 $ 51 Bats 26 73 79 Shoes 47 Uniforms 51 92 37 96 37 Compute the lower of...
-
How can the machine epsilon be employed to formulate a stopping criterion s for your program? Provide an example.
-
A proton and an electron form two corners of an equilateral triangle of side length 2.0 x 10-6 m. What is the magnitude of the net electric field these two particles produce at the third corner?
-
(a) What total (excess) charge q must the disk in Figure have for the electric field on the surface of the disk at its center to have magnitude 3.0 x 106 N/C, the E value at which air breaks down...
-
Calydon Corporation manufactures and distributes wooden baseball bats. This is a seasonal business with a large portion of its sales occurring in late winter and early spring. The production schedule...
-
Based on NLRB opinions on Social Media , analyze the approach a company should take with approaching a Social Media policy. Describe the pros and cons of having a social media policy and determine...
-
In consideration of your evolving understanding of Dr. Deming's System of Profound Knowledge, is process stability needed before a process can be improved? In consideration of your evolving...
-
Designing a plan for retaining top talent and developing a succession plan: To design a plan for retaining top talent and developing a succession plan for a job, you can follow these steps: Identify...
-
According to retention management strategies complete the questions below: Questions: (In the case of the Society for Human Resource Management) why does retention start early with an employee? How...
-
Design a Recruitment Plan Great news! Just as expected, XYZ Company is growing! Sales are at record high levels, and the CEO has approved hiring five new team members. Your job is to design...
-
An electric power plant uses energy from burning coal to generate steam at \(450^{\circ} \mathrm{C}\). The plant is cooled by \(20^{\circ} \mathrm{C}\) water from a nearby river. If burning coal...
-
31. What is the income that can be received over 15 years from $500,000 earning 6% annually? 32. What is the semiannual payment required to retire $50,000 in debt over 5 years at 8% compounded...
-
This exercise proves the Chain Rule without the special assumption made in the text. For any number b, define a new function (a) Show that if we define F(b) = '(b), then F is continuous at u = b. (b)...
-
You make tea with 0.250 kg of 85.0C water and let it cool to room temperature (20.0C) before drinking it. (a) Calculate the entropy change of the water while it cools. (b) The cooling process is...
-
Three moles of an ideal gas undergo a reversible isothermal compression at 20.0oC. During this compression, 1850 J of work is done on the gas. What is the change of entropy of the gas?
-
What is the change in entropy of 0.130 kg of helium gas at the normal boiling point of helium when it all condenses isothermally to 1.00 L of liquid helium? (Hint: See Table 17.4 in Section 17.6.)
-
Golden Gate Mining Co. (GGMC) currently has a gold mine operating in Canada and is looking to purchase another gold mine in Peru, operating as Inca Gold Inc. (ICI). You currently work for the CFO at...
-
Cokolo Inc. has entered into the following two derivatives contracts: Purchased put options on 1,000 Trenton Co. shares that have an exercise price of $52 per share and expire in 60 days Negotiated a...
-
As a financial manager (consultant), undertake an investigation on your allocated company to evaluate its strategic financial position. Prepare a business report for the board of directors of the...

Study smarter with the SolutionInn App


