Question: In light of your answer to Problem 11.61, explain why one of the following isomers undergoes E2 reaction approximately 100 times as fast as the
In light of your answer to Problem 11.61, explain why one of the following isomers undergoes E2 reaction approximately 100 times as fast as the other. Which isomer is more reactive, andwhy?
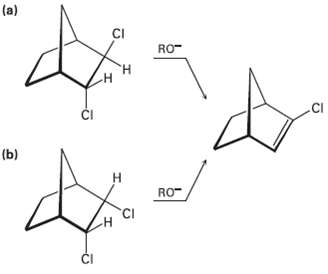
(a) CI RO- .CI I (b) RO- CI I
Step by Step Solution
3.34 Rating (175 Votes )
There are 3 Steps involved in it
H N H H A B We concluded in Problem 1161 that E2 elimination i... View full answer

Get step-by-step solutions from verified subject matter experts
Document Format (1 attachment)
22-C-O-E-R (155).docx
120 KBs Word File


