In planning the synthesis of one compound from another, it?s just as important to know what not
Question:
In planning the synthesis of one compound from another, it?s just as important to know what not to do as to know what to do. The following reactions all have serious drawbacks to them. Explain the potential problems of each.
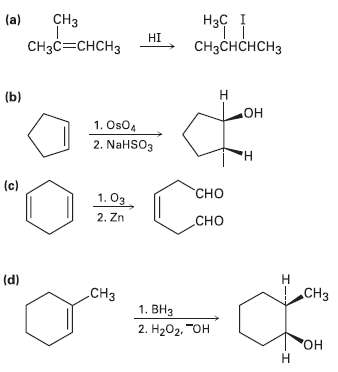
Transcribed Image Text:
нsf CHз (a) Наҫ HI снас—снсHз снаснснсHз Н (b) он 1. Os04 2. NaHSO3 н (c) "Сно 1. Оз 2. Zn Сно Н (d) CHз .CНз 1. ВНз 2. Н202, "Он "Он Н
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (10 reviews)
a Addition of HI occurs with Markovnikov regiochemistry iodine ad...View the full answer

Answered By

Susan Juma
I'm available and reachable 24/7. I have high experience in helping students with their assignments, proposals, and dissertations. Most importantly, I'm a professional accountant and I can handle all kinds of accounting and finance problems.
4.40+
15+ Reviews
45+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
In planning synthesis, it?s as important to know what not do as to know what to do. As written, the following reaction schemes have flaws in them. What is wrong with each? (a) CN CN 1. CH3CH,COCI,...
-
Explain why strategic planning is important to all managers.
-
It is not important to know when cash is received and when payment is made. Comment.
-
Ratio Computation and Analysis; Liquidity) as loan analyst for Madison Bank, you have been presented the following information. Each of these companies has requested a loan of $50,000 for 6 months...
-
Over the past month, Bob's bowling score mean was 182 with a standard deviation of 9.1. His bowling partner Cedric's mean was 152 with a standard deviation of 7.6. Which bowler is more consistent in...
-
Lorena is a golfer who wants to investigate whether she successfully makes more than 60% of her 10-foot putts. a. Define (in words) the relevant parameter of interest for Lorena. b. State (in words...
-
In a study measuring households' familiarity with downloading pictures from the Internet, the following results were obtained $(1=$ not at all familiar, $7=$ very familiar $)$. Level of Familiarity...
-
Under the UCC, which party, Golden Years or Star Bank, must bear the loss in this situation? Why? Nancy Mahar was the office manager at Golden Years Nursing Home, Inc. She was given a signature stamp...
-
On January 1, JKR Shop had $550,000 of beginning inventory at cost. In the first quarter of the year, it purchased $1,690,000 of merchandise, returned $24,100, and paid freight charges of $38,600 on...
-
Sketch vo for the network of Fig. 2.170 and determine the dc voltage available. Ideal diodes 170 V 2.2 k -Vo + 2.2 k -170 V 2.2 k2
-
SimmonsSmith reaction of cyclohexane with di iodomethane gives a single Cyclopropane product, but the analogous reaction of cyclohexane with 1, 1-diiodoethane gives (in low yield) a mixture of two...
-
Which of the following alcohols could not be made selectively by hydroboration oxidation of an alkene? Explain. (a) ( , CH3CH2CH2CHCH3 (CH3)2HCICH)2 CH (c) (d) .CH " H.
-
1. Use the following information to complete the Adjustments columns of the work sheet. a. Depreciation on equipment, $3 d. Supplies available at December 31, $15 b. Accrued salaries, $6 e. Expired...
-
Paula is a retailer operating in Toowoomba and she received the following amounts during the current tax year ended 30 June: Paula received $320 000 from the sale of an investment property she had...
-
1) Pursuant to a verbal contract, Pamela was hired and worked from July 1 as a lifeguard for the outdoor pool at the Midnight Sun Resort and Spa in Northern Alberta. When she arrived for work on the...
-
Elizabeth, a resident, carries on a retail business selling health foods to the public. Up to 30 April o the current tax year she operated her business from her own premises. On 30 April she sold her...
-
find the value of X x+30 2x+1 = 3
-
Section 107 (3) of the Industrial and Labour Relations Act, Cap 269 of the Laws of Zambia provides that; no employer or other person shall take part in a lockout and no employee, trade union or other...
-
Consider the following transfer function: \[G(s)=\frac{1}{s^{2}+10 s+100}\] (a) Express the transfer function in terms of a constant factor and a complex pole factor. (b) Draw the Bode plots for the...
-
Under what conditions is the following SQL statement valid?
-
Which molecules have dipoledipole forces? (a) CO 2 (b) CH 2 Cl 2 (c) CH 4
-
(a) According to the mechanism of the reaction shown in Eq. 10.24, what would be the absolute configuration of the alkyl chloride obtained from the reaction of thionyl chloride. With (S)...
-
Suggest conditions for carrying out each of the following conversions to yields a product that is as free of isomers as possible. (a) (CH3)2CH(CH2)OH (CH3)3CH(CH2)4CI (b) Br OH
-
Suggest conditions for carrying out each of the following conversions to yields a product that is as free of isomers as possible. (a) (CH3)2CH(CH2)OH (CH3)3CH(CH2)4CI (b) Br OH
-
The fact that restrictive covenants are widely used does not mean, as NanoMech, Inc. v. Suresh shows, that they are always enforceable. NanoMech, Inc. v. Suresh 777 F.3d 1020 (8th Cir. 2015) OPINION...
-
What are the fundamental distinctions between prokaryotic organisms and their eukaryotic counterparts, particularly concerning cellular structure and organizational complexity?
-
What are the structural and biochemical features of prokaryotic cell membranes and cell walls, and how do these components contribute to cellular integrity, homeostasis, and environmental...

Study smarter with the SolutionInn App


