Which of the following alcohols could not be made selectively by hydroboration oxidation of an alkene? Explain.
Question:
Which of the following alcohols could not be made selectively by hydroboration oxidation of an alkene? Explain.
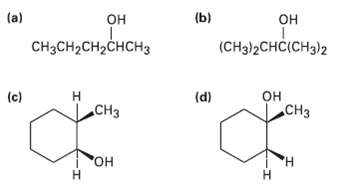
Transcribed Image Text:
(a) (ы он снонененон, CH3CH2CH2CHCH3 (CH3)2СHCICHз)2 он CHз Н (c) (d) .CHз "Он Н H. Н т т
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (15 reviews)
a This alcohol cant be synthesized selectively by hydroborationoxid...View the full answer

Answered By

Fahmin Arakkal
Tutoring and Contributing expert question and answers to teachers and students.
Primarily oversees the Heat and Mass Transfer contents presented on websites and blogs.
Responsible for Creating, Editing, Updating all contents related Chemical Engineering in
latex language
4.40+
8+ Reviews
22+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Which of the following numbers could not be probabilities, and why? a. 125% b. 0.74 c. 0.001 d. 5.61 e. 150%
-
Which of the following alcohols can be synthesizecl relatively free of constitutional isomers and diastereomers by oxymercuration-reduction? Explain. H,C CH,CH2CH CH2CH CH H,C H,C
-
Which of the following constraints are not linear or cannot be included as a constraint in a linear programmingproblem? a. 2X1 + X2 3X3 50 b. 2x V60 c. 4x1-x2 = 75 d. 3x1 + 2x2-3x30.9 3x7+7x2 < 45 e.
-
Northland Corporation is a small information-systems consulting firm that specializes in helping companies implement standard sales-management software. The market for Northalndss services is very...
-
Spirit Airlines kept track of the number of empty seats on flight 308 (DEN-DTW) for 10 consecutive trips on each weekday except Friday. (a) Sort the data for each day. (b) Find the mean, median,...
-
The copper-silver phase diagram is shown in Figure 11-30. Copper has a higher melting point than silver. Refer to the silver-rich solid phase as gamma (γ) and the copper-rich solid phase...
-
Data set: Ratings from 1 (lowest) to 10 (highest) provided by 36 people after taste testing a new flavor of ice cream. Construct a frequency distribution and a relative frequency histogram for the...
-
Briefly describe the elements of a business case for a new information systems project under the SDLC methodology.
-
(1) A car is traveling at a constant speed of 5 m/s. If the snow is falling at 4.1 m/s with an angle of 36 degrees to the vertical line toward the windshield, determine the velocity of the snow as...
-
Let total market demand for labor be represented by ED = 1,000 - 50w where ED is total employment and w is the hourly wage. a. What is the market clearing wage when total labor supply is represented...
-
In planning the synthesis of one compound from another, it?s just as important to know what not to do as to know what to do. The following reactions all have serious drawbacks to them. Explain the...
-
Predict the products of the following reactions. Don?t worry about the size of the molecule; concentrate on the functional groups. Br2 A? C Br B? CH3 1. OsO4 2. NaHSO3 C? 1. BH3, THE D? 2. 20. " ...
-
A scuba diver sets off a camera flash at depth h in water with refractive index n. Show that light emerges from the waters surface through a circle of diameter 2h/ (n 2 - 1).
-
A climber on Mount Everest is 6000 meters from the start of his trail and at elevation 8000 meters above sea level. At x meters from the start, the elevation of the trail is h (x) meters above sea...
-
ABC Limited is looking to acquire a new equipment for its project that will last for five years. The required rate of return of the project is 10% per annum. ABC can borrow at 9% per annum and buy...
-
4. Subtract the following the Hex numbers: Result is = 67h 2Ah
-
Choose a brand and scan the internet (blogs, website reviews etc), explain in detail the top 5 topics being discussed, indicate if the brands are positive or negative and explain in detail some of...
-
The Consumer Price Index (CPI) is a series of price indices calculated by the U.S. Bureau of Labor Statistics (BLS) per month. Suppose we know the average CPI was 259 in December 2019. After years,...
-
It is April 7, 2022. The quoted price of a U.S. government bond with a 6% per annum coupon (paid semiannually) is 120-00. The bond matures on July 27, 2033. What is the cash price? How does your...
-
Write an SQL statement to display all data on products having a QuantityOnHand greater than 0.
-
The VSEPR model is useful in predicting bond angles for many compounds. However, as we have seen, other factors (such as type of bond and atomic radii) may also influence bond angles. Consider that...
-
Give the curved-arrow mechanism for the reaction in Problem 10.la. In each step, identify all Bronsted acids and bases, all electrophiles and nucleophiles, and all leaving groups.
-
Give the curved-arrow mechanism for the reaction in Problem 10.la. In each step, identify all Bronsted acids and bases, all electrophiles and nucleophiles, and all leaving groups.
-
Considering the organic compound, classify each of the following transformation, some of which may be unfamiliar, as an oxidation, a reduction, or neither, For those are oxidation or tell how may...
-
Golden Gate Mining Co. (GGMC) currently has a gold mine operating in Canada and is looking to purchase another gold mine in Peru, operating as Inca Gold Inc. (ICI). You currently work for the CFO at...
-
Cokolo Inc. has entered into the following two derivatives contracts: Purchased put options on 1,000 Trenton Co. shares that have an exercise price of $52 per share and expire in 60 days Negotiated a...
-
As a financial manager (consultant), undertake an investigation on your allocated company to evaluate its strategic financial position. Prepare a business report for the board of directors of the...

Study smarter with the SolutionInn App


