In the iodoform reaction, a triiodomethyl ketone reacts with aqueous NaOH to yield a carboxylate ion and
Question:
In the iodoform reaction, a triiodomethyl ketone reacts with aqueous NaOH to yield a carboxylate ion and iodoform (triiodomethane). Propose a mechanism for thisreaction.
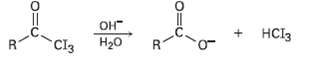
Transcribed Image Text:
OH H20 Cl3 HCI3 R.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 62% (16 reviews)
This is a nucleophilic acyl substitution reaction who...View the full answer

Answered By

Joseph Mwaura
I have been teaching college students in various subjects for 9 years now. Besides, I have been tutoring online with several tutoring companies from 2010 to date. The 9 years of experience as a tutor has enabled me to develop multiple tutoring skills and see thousands of students excel in their education and in life after school which gives me much pleasure. I have assisted students in essay writing and in doing academic research and this has helped me be well versed with the various writing styles such as APA, MLA, Chicago/ Turabian, Harvard. I am always ready to handle work at any hour and in any way as students specify. In my tutoring journey, excellence has always been my guiding standard.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Organic Chemistry questions
-
Propose a mechanism for the reaction of cyclohexyl methyl ketone with excess bromine in the presence of sodium hydroxide.
-
Propose a mechanism that shows why p-chlorotoluene reacts with sodium hydroxide at 350 C to give a mixture of p-cresol and m-cresol.
-
Propose a mechanism for the reaction of acetyl chloride with phenylmagnesium bromide to give 1,1-diphenylethanol. OH (1) ether solvent CH3-C CI 2 (2) H 0 acetyl chloride phenylmagnesium bromide...
-
Consider the following function. def f(x): a = [] while x > 0: a.append(x) f(x-1) A new object of type list is created for each recursive invocation of f. True or False
-
Explain how blogs and social media networks can be used by businesses and report writers.
-
Classify each of the frames as statically determinate or indeterminate. If indeterminate, specify the degree of indeterminacy. All internal joints are fixed connected. a. b. c. d.
-
You have been meeting with a potential new customer regularly for three months. She likes the product but finally admits a loyalty to the existing supplier. The buyer says, I have known Judith...
-
Refer to the previous question. Suppose that each of the audit activities can be crashed by the amounts indicated in the following table. a. What is the earliest the audit could be completed and what...
-
RH (a) Alternative 1 (b) Alternative 2 Figure 1. Schematic layout of alternative engine construction. Table 1 Specifications of the Whoop-de-do 5000 engine. Engine Specifications Capacity Bore Stroke...
-
Determine Zi Zo and Av for the network of Fig. 8.84. +20 V aSS 12 mA 3 91 = 45 k Z 10 MS2
-
Epoxy adhesives are prepared in two steps. SN2 reaction of the disodium salt of bisphenol A with epichiorohydrin forms a ?prepolymer,? which is then ?cured? by treatment with a triamine such as H 2...
-
Draw structures for the enol tautomers of the following compounds: (a) Cyclopentanone (b) Methyl thioacetate (c) Ethyl acetate (d) Propanal (e) Acetic acid (f) Phenyl acetone
-
Refer to Exercise 15.8. a. Describe how the four fertilizer placement rate combinations are randomly assigned to the rows and columns in the farm plot. b. Compute the relative efficiency of the Latin...
-
Find the consumer surplus, when the monopolists sets one price given the following inverse demand, marginal revenue, and marginal cost functions: Inverse Demand: P = 220- 5Q Marginal Revenue =...
-
Generate a question related to ethical perspectives in persuasion and social influence, after reading the assigned readings. You can choose a topic such as honesty versus dishonesty and its impact on...
-
Globalization demands a single set of high quality international accounting standards. List the elements of High Quality Standards and explain the two major boards that sets standards.
-
Provide an explanation of the potential cost and time for each assessment (such as the expense of the Hogan Test and the one hour required to complete the exam) as well as the return on investment...
-
Identify 4 business risks of Integral Diagnostics Limited (IDX) that will increase the risk of material misstatement (specifically inherent risk) at the assertion level for classes of transactions or...
-
Table 3.5 shows ringer percentages for Mary Ann Peninger at the 2000 Womens world horseshoe championships, when she threw first and when she threw second. For example, in the second game, she threw...
-
A liquid flows upward through a valve situated in a vertical pipe. Calculate the differential pressure (kPa) between points A and B. The mean velocity of the flow is 4.1 m/s. The specific gravity of...
-
Explain the difference between hybrid atomic orbitals in valence bond theory and LCAO molecular orbitals in MO theory.
-
Explain why The methyl group in the following compound has an unusual chemical shift of (- 1.61), about 4 ppm lower than the chemical shift of a typical allylic methyl group. : Na sodium salt of...
-
Explain why The methyl group in the following compound has an unusual chemical shift of (- 1.61), about 4 ppm lower than the chemical shift of a typical allylic methyl group. : Na sodium salt of...
-
Within each set, which compound should show NMR absorptions with the greater chemical shifts? Explain your choices. (1) (2)
-
E3-20 (Algo) Analyzing, Recording, and Evaluating the Effects of Income Statement Transactions [LO 3-3, LO 3-4, LO 3-5] [The following information applies to the questions displayed below.] MyBnB...
-
Required information [The following information applies to the questions displayed below.] The following data reports on the July production activities of the Molding department at Ash Company....
-
Question 1 Vieux Enterprise's total overhead costs at various levels of activity follow: Month Machine- Hours Total Overhead Costs April 70,000 $198,000 May 100,000 270,000 June 80,000 222,000 July...

Study smarter with the SolutionInn App


